21-22 June 2018
LanguageCN/EN
SCRA 2018 HighlightsWhy Attend
Who Shall Attend
Simultaneous interpretation is available in Chinese and English. The agenda is updated on 27 March 2018. We apologize for any inconvenience due to the change of agenda.
|
14:00-18:00 20th June |
Registration You are encouraged to complete the check-in procedure on the afternoon of 20th June to receive delegate card/meeting materials for easy access and early review. |
|
| Day 1 | Topics | Speaker |
| 8:15-8:45 | Registration | |
| 8:45-9:00 | Welcome | Mr. Rongqin Zhang, Chairman, CACPQSP |
| 9:00-9:40 | Management trend of Chinese cosmetics and cosmetic ingredient in 2018 | Mr. Liubin Qi, Director, Department of Drug and Cosmetic Registration, CNDA, State Administration for Market Regulation |
| 9:40-10:00 | Panel Discussion | |
| 10:00-10:20 | Tea and Coffee | |
| 10:20-11:00 | Supervision of Chinese imported & exported cosmetics | Mr. Yue Yu, Head of Food Safety Supervision Department, Hangzhou Customs |
| 11:00-11:10 | Panel Discussion | |
| 11:10-11:50 | Record-keeping requirements of imported non-special use cosmetics product in Zhejiang free trade area | Ms. Juxiang Zheng, Division Chief, Zhejiang Food and Drug Administration |
| 11:50-12:00 | Panel Discussion | |
| 12:00-13:00 | Lunch | |
| 13:00-13:40 | Technical review requirements of imported non-special use cosmetic in free trade area | Mr. Liang Xu, Director of Beijing Daily-Use Chemicals Institute |
| 13:40-14:20 | Technical review requirements of special use cosmetic in China | Ms. Hongwei Zhang, Director, National Institute of Environmental Health Chinese Center for Disease Control and Prevention |
| 14:20-15:10 | Evaluation method on cosmetic efficacy | Ms Yimei Tan, Deputy Director of Skin and Cosmetic Laboratory, Shanghai Dermatology Hospital |
| 15:10-15:30 | Tea and Coffee | |
| 15:30-16:10 | Alternative method to evaluate the consistency of efficacy on cosmetic ingredients | Mr. Shujun Cheng, Laboratory Chief, Guangdong Entry-Exit Inspection and Quarantine Technical Center |
| 16:10-16:50 | Panel Discussion | |
| Day 1 Concluded | ||
| Day 2 | ||
| 9:00-9:40 | Latest development of EU cosmetic regulation | Mr. Gerald Renner, Director Technical Regulatory & International Affairs, Cosmetics Europe |
| 9:40-10:20 | Cosmetic management regulation in the US | Ms. Sharon Blinkoff, Chair of Legal Committee, ICMAD |
| 10:20-10:40 | Tea and Coffee | |
| 10:40-11:20 | How do you manage to launch quasi-drug & cosmetics in Japan | Dr. Masato Hatao, Director for Safety & Science, Japan Cosmetic Industry Association |
| 11:20-11:50 | Panel discussion | |
| 11:50-13:00 | Lunch | |
| 13:00-13:40 | Regulatory compliance on cosmetic raw materials in China | Ms. Chloe Wang, Technical Manager of Cosmetics Regulatory Affairs from CIRS Technology Group |
| 13:40-14:20 | Management requirements on cosmetic raw materials in EU | Mr. Gerald Renner, Director Technical Regulatory & International Affairs, Cosmetics Europe |
| 14:20-15:00 | Management requirements on cosmetic raw materials in the US | Mr. Craig R. Weiss, Chair of Technical and Regulatory Committee, ICMAD |
| 15:00-15:20 | Tea and Coffee | |
| 15:20-16:00 | Introduction of IFS HPC standard | Ms. Helga Barrios, Senior Technical Project Manager, IFS Management GmbH |
| 16:00-16:30 | Panel Discussion | |
| Summit Concluded | ||

China Association for Consumer Products Quality and Safety Promotion (CACPQSP), established at the approval of Ministry of Civil Affairs of the People's Republic of China, is under the charge of State Administration for Quality Supervision and Inspection and Quarantine (AQSIQ). It is also a national non-profit organization with independent legal qualification, the basic objective of which is to strengthen social supervision, provide consultation service and build a bridge between central government and enterprises, thereby push the comprehensive quality of consumer products, ensure the consumption safety in China and enhance our profile at home and abroad.

Chemical Inspection and Regulation Service(CIRS) is an independent firm providing professional chemical regulatory consulting, certification and testing services to a variety of industries such as the chemical industry, consumer product industry, scientific research institutions and industry associations. We are dedicated to providing values chemical compliance services, tailored solutions and original information to help our clients gain competitive advantages by reducing business risks associated with regulatory affairs and removing barriers to entry.
With its headquarter based in Hangzhou of China and offices or laboratories located in Ireland, Beijing, Nanjing, Ningbo and Wenzhou, CIRS utilizes its technical expertise, various resources and global network to provide comprehensive compliance services such as chemical notification, GHS compliance services, pesticides and biocides registration, cosmetics and new cosmetic ingredient registration, chemical testing, regulatory update monitoring and training services.
| For New Attendee (VAT inclusive) | Previous Attendee (VAT inclusive) | |
| Early-bird Rate(Before 1 May) | 3400RMB/540USD/440EUR per delegate | 3200RMB/510USD/420EUR per delegate |
| Standard Rate (After 1 May) | 3600RMB/570USD/460EUR per delegate | 3400RMB/540USD/440EUR per delegate |
*Please note registration fee include two luncheons, and accommodation is borne by delegates.
How to register
Ms.Susan Shen
Tel:0086 571 8720 6574
Email: susan@cirs-group.com
Address:11/F Building 1, Dongguan Hi-Tech Park, 288 Qiuyi Road, Binjiang District, Hangzhou, China, 310052
Payment Approaches
1. Bank transfer
China
| Bank information | EN | CN |
| Account Name | Hangzhou REACH technology Group Co,.Ltd. | 杭州瑞旭科技集团有限公司 |
| Bank Name | ICBC Hangzhou, Qianjiang Sub-branch | 工商银行杭州钱江支行 |
| Account No. | 1202021419900146022 | |
| Swift Code | ICBKCNBJZJP | |
| Row Number (Bulk Payment Number) | 102331002149 | |
Europe
| Beneficiary Name | Chemical Inspection and Regulation Service Ltd. |
| Beneficiary Address | Singleton House, Drogheda, Co. Louth |
| Bank Name | Bank of Ireland |
| Bank Address | Wilton, Cork, Republic of Ireland |
| Sort Code | 902805 |
| Account # | 90404884 |
| BIC/Swift | BOFIIE2D |
| IBAN | IE15 BOFI 9028 0590 4048 84 |
Please contact Susan at susan@cirs-group.com before transfer.
2. Cash
We also accept cash as payment when you attend this event. Receipt will be given during the summit.
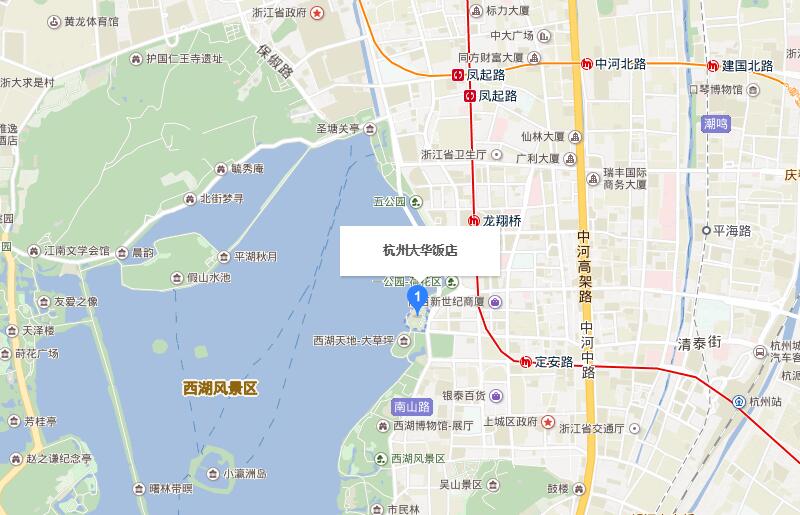
The Dahua Hotel is located just a few steps away from the charming West Lake in central Hangzhou. The hotel featuring classic courtyard-style and once receiving Chairman Mao makes it more attractive.
Address:
No.171 Nanshan Road, Shangcheng District, Hangzhou
Tel:
86-0571-87181888
The closest Metro Station is Dingan Road Station/ Longxiangqiao Station. You can arrive at the station by M1 of Hangzhou Metro.
Nearby Destinations
- West Lake (0.5 km)
- Westlake Intime Shopping Mall (1.0 km)
- SouthernSong Imperial Street (1.4 km)
- Hangzhou Xiaoshan International Airport(HGH)(30KM)