As of March 31, 2026, a total of 20 Foods for Special Medical Purposes (FSMPs) were approved in the first quarter of 2026, bringing the cumulative number of approved FSMPs in China to 310.
Table 1. 20 FSMPs Approved in the First Quarter of 2026
Register No. | Product Name | Applicant |
|---|---|---|
国食注字TY20260001 | 若兴 | Shandong Ruoyao |
国食注字TY20260004 | 卡焕® | Masisa (Taizhou) |
国食注字TY20260005 | 舒益素太® | Aiyounuo |
国食注字TY20260006 | 立优荃® | Shenzhen Jielikang |
国食注字TY20260007 | 纽先 | Shenzhen Jielikang |
国食注字TY20260008 | 力优宜佳 | Wuxi Licheng |
国食注字TY20260009 | 特康优素® | Tekang Pharmaceutical |
国食注字TY20260010 | 小优达力® | Aiyounuo |
国食注字TY20260011 | 水达素® | Zhongte |
国食注字TY20260012 | 质沛® | Tekang |
国食注字TY20260013 | 恩立安 | Zhongen (Tianjin) Pharmaceutical |
国食注字TY20260014 | 特怡全 | Yichang Humanwell |
国食注字TY20260015 | 卡特泰 | Yunnan Institute of Materia Medica |
国食注字TY20260016 | 特益力 | Shandong Ruoyao |
国食注字TY20260017 | 瑞葆安® | Inner Mongolia Tekangrui |
国食注字TY20260018 | 玛士素® | Masisa (Taizhou) |
国食注字TY20260019 | 冬泽全瑞 | Masisa (Taizhou) |
国食注字TY20260020 | 特维聚® | Hunan Shengmingyuan |
国食注字TY20260021 | 特维坦® | Hunan Shengmingyuan |
国食注字TY20260022 | 欣全膳® | Chenxin Pharmaceutical |
CIRS conducted a comprehensive statistical summary of the 20 FSMPs and carried out multi-dimensional analyses to provide insights and references for enterprises.
General Situation of FSMPs in the First Quarter of 2026
In the Q1 of 2026, all the approved 20 FSMPs were domestic products, accounting for 100%. In terms of product dosage forms, powder products were the most common, with a total of 16, accounting for 80%; liquid products numbered 4, accounting for 20%.
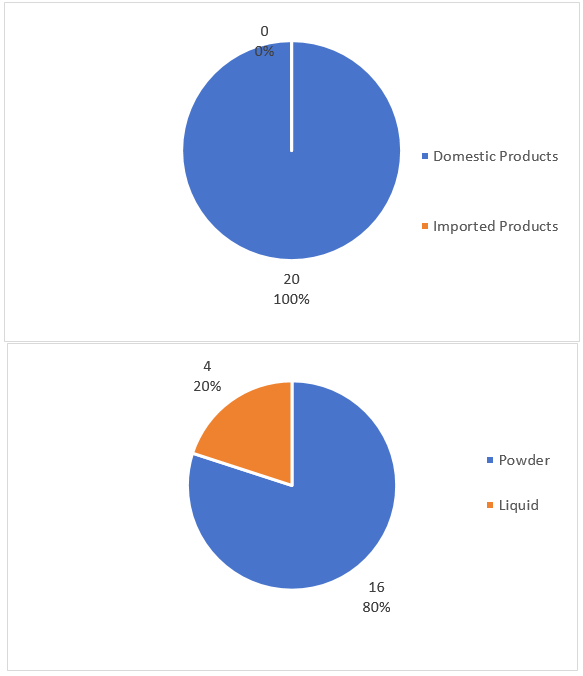
Figure 1. General Situation of FSMPs in Q1 of 2026
Number of FSMP Registrations in Q1 of 2026 (by Approval Time)
The distribution of registration approval times for FSMPs in Q1 of 2026 is shown in Figure 2. From the chart, it can be seen that, overall, the number of approvals in January and February was roughly the same. There is currently no data for March, likely due to delays in information release. Approvals granted in March are expected to be announced in April.
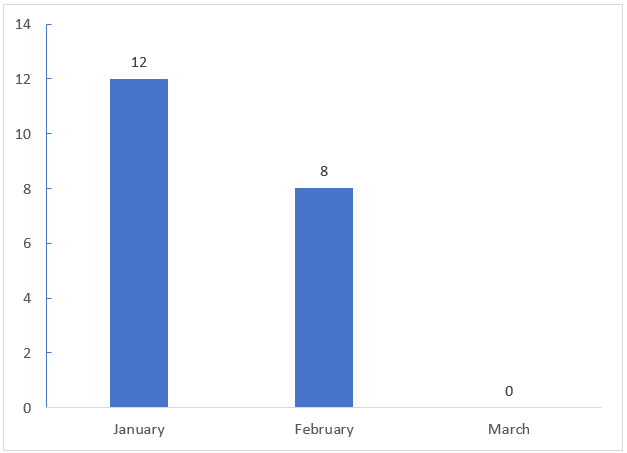
Figure 2. Monthly Number of FSMP Registrations Approved in Q1 of 2026
3. Number of FSMP Registrations Approved in Q1 of 2026 (by Product Category)
As shown in Figure 3 below, among the FSMPs approved for registration in Q1 of 2026, the number of complete nutrition formula foods was the highest, with a total of 13, accounting for 65% of the total approved number (among them, there were five protein components, four electrolyte formulations, two fat components, one liquid formulation, and one carbohydrate component); secondly, there were six total nutrition formulas, accounting for 30% of the total approvals; the remaining one was a formula for amino acid metabolism disorders.
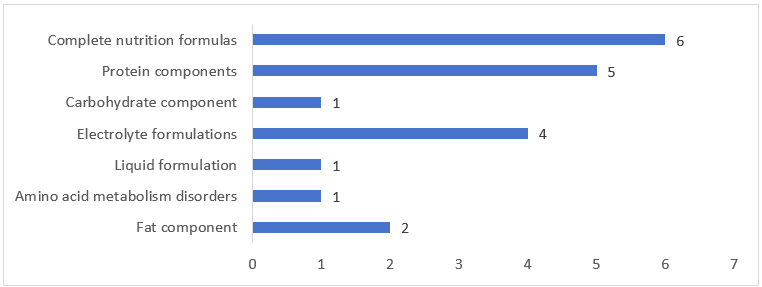
Figure 3. Number of Registrations Approved for Different Categories of FSMPs in Q1 of 2026
Number of FSMP Registrations Approved in Q1 of 2026 (by Target Population)
As shown in Figure 4 below, among the FSMPs approved for registration in Q1 of 2026, the largest number of products were intended for people aged ten years and above, totaling 14. This was followed by products for individuals aged one year and above (3 products), those aged 1–10 years (2 products), and infants aged 0–12 months (1 product). Overall, the distribution reflects coverage across the entire life cycle while addressing key population groups, with a particularly significant proportion of products targeting adults.
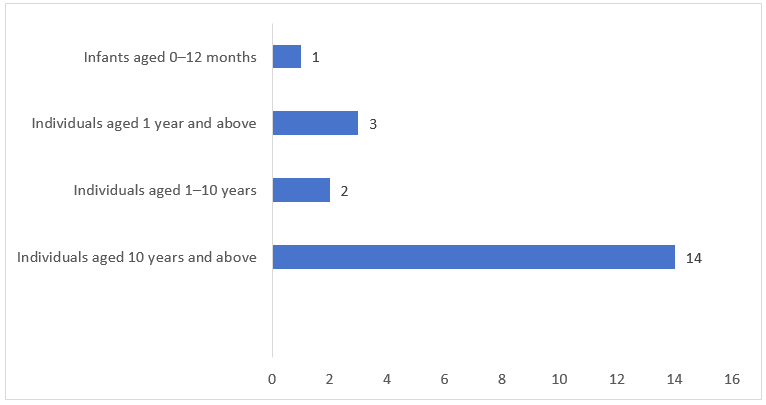
Figure 4. Number of Registrations Approved for FSMPs by Target Population in Q1 of 2026
Number of FSMP Registrations Approved in Q1 of 2026 (by Province/Region)
The 20 special medical purpose foods approved for registration in Q1 of 2026 came from 10 different provinces and regions. Shandong province, Jiangsu province, and Jiangxi province ranked top three with 5, 4, and 3 products respectively, accounting for 60% of the total approved number. It is worth noting that both Yunnan and Inner Mongolia obtained their first approvals for FSMPs in the first quarter of 2026, marking a breakthrough from zero and indicating that their industry development has begun to take shape. For specific provincial distribution and data, see Figure 5 below.
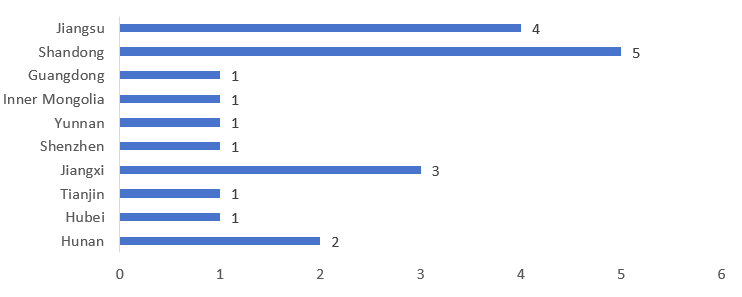
Figure 5. Number of Registrations Approved for FSMPs by Province/Region in the Q1 of 2026
Number of FSMP Registrations Approved in Q1 of 2026 (by Applicant Unit)
The 20 FSMPs approved for registration in Q1 of 2026 involved 14 applicant units. As shown in Figure 6 below, six companies, including Aiyounuo, each obtained approvals for two FSMPs, while the remaining eight companies each received approval for one FSMP
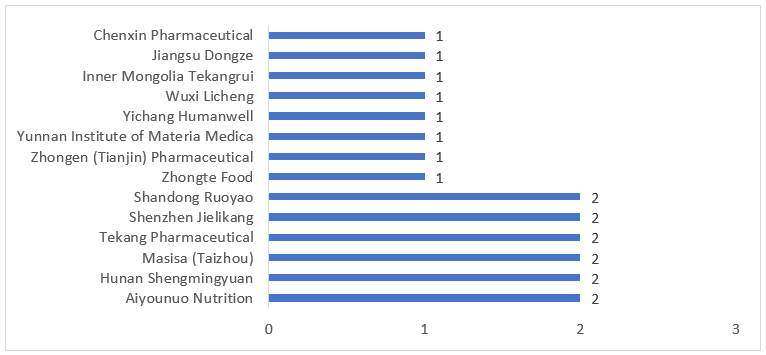
Figure 6. Top Companies by Number of FSMP Registrations Approved in Q1 of 2026
CIRS Analysis
At present, FSMPs have achieved preliminary coverage across major categories; however, there remain significant product gaps in certain sub-segments, such as specific complete nutrition formulas and formulas for amino acid metabolism disorders. With the continuous improvement of relevant technical guidelines and the gradual implementation of priority review policies, FSMPs targeting special medical needs – such as rare diseases and chronic conditions – are expected to accelerate in approval and market entry in the future.
About CIRS
CIRS Group has a professional registration team and extensive project experience. Whether your product is imported or domestically produced, our experienced technical experts can provide one-stop solutions to support efficient compliance. Feel free to contact or visit us for further consultation.
If you need any assistance or have any questions, please get in touch with us via service@cirs-group.com.
Notes:
1. Data Source: Special Food Information Inquiry Platform.
2. There may be a delay in the release of data from the Special Food Information Inquiry Platform. The data in this article is for reference only, and the actual situation should be based on official announcements.


