India's Regulations on Imported Chemicals took effect on October 1, 2023. Originally, the regulation was set to take effect on July 1, 2023.
On September 30, 2023, the Central Board of Indirect Taxes and Customs in India issued Circular No.23/2023, which made the following modifications to sections (4.1) and (4.2) of Circular No.15/2023:
4.1 Commodities imported under Chapters 28, 29, 32, heading 3808, and Chapter 39, are required to provide additional information at the time of filing import declarations as follows:
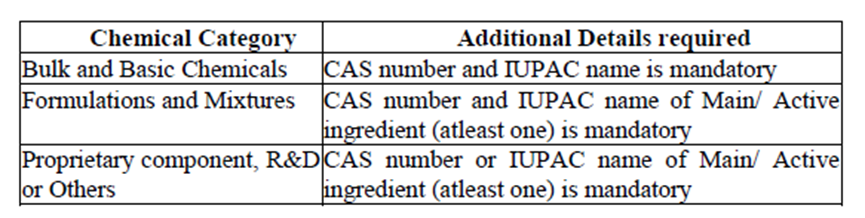
IUPAC = universally-recognized authority on chemical nomenclature and terminology.
4.2 If the supplier does not share information for even one ingredient with the importer due to confidentiality, the importer must provide a self-undertaking to declare the non-availability of the CAS number and IUPAC details.
Details of constituents in the Bill of Entry will be printed as a Masked field in the Bill of Entry. These additional identifiers will be mandatory for all bills of entry filed on or after October 15, 2023.
This change aims to set higher standards for chemical imports to India, enhance the transparency for imported goods, ensure chemical safety and traceability, and improve customs assessment efficiency. Authorities will release notifications to provide guidance and encourage stakeholders to submit feedback for timely resolutions.
If you need any assistance or have any questions, please contact us via service@cirs-group.com.
Further Information

