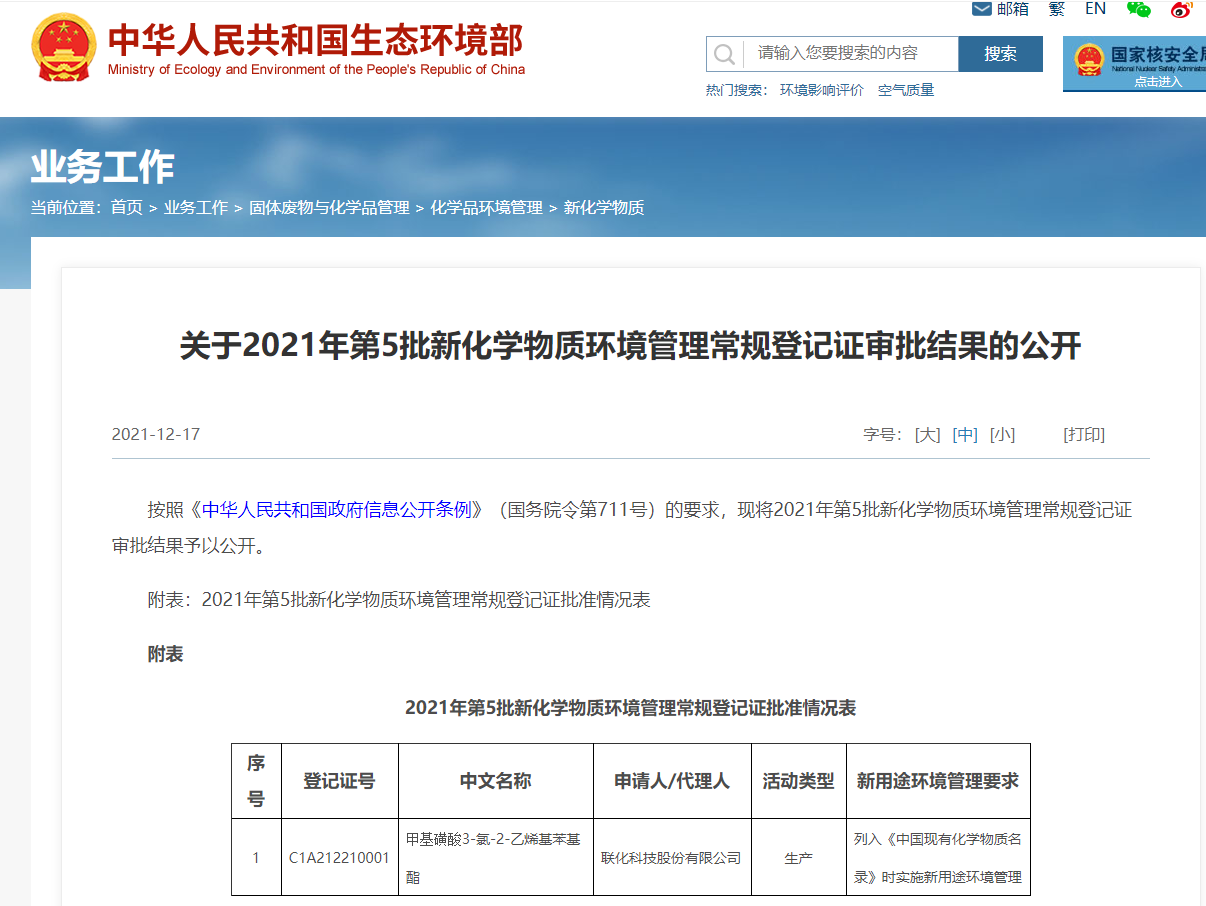
The Provisions on Environmental Administration Registration of New Chemical Substances (MEE Order No. 12) has been implemented for over a year. It has attracted extensive attentions from related enterprise. However, as the data requirements for regular registrations under MEE Order No. 12 are higher than that of MEP Order No. 7, only 11 batches of approved regular/simplified registrations, which includes 38 simplified registrations and 1 regular registration, have been notified.
As a technical supporter, CIRS successfully helped clients complete the first regular registration under MEE Order No. 12. Below are the registration experience from CIRS:
1. Analyse Data Gap and Set Reasonable Project Expectations
Before regular registration, enterprises should make sure:
- Whether the substances are special substances: such as substances not able to test, inorganic substances, substances that will decompose when in contact with water/light;
- Whether the substances are used only as pesticide, pharmaceutical or veterinary intermediates: If the substances are registered only as pesticide, pharmaceutical or veterinary intermediates, then enterprises shall submit basic information only when processing regular registrations.
The special data requirements, such as reproductive toxicity of two generations, chronic toxicity, carcinogenicity, etc. requires higher experiment cost and longer experimental periods, which will seriously restrict the registration process. For this reason, generally speaking, if the substances subject to registration are special substances, especially special substances registered only as pesticide, pharmaceutical or veterinary intermediates, it is expected that the regular registration process will be smoother than that of the general substances.
2. Reasonably Plan a Test and Judge the Properties of Substance (PB)
When processing regular registrations, substances that are not persistent (P) and Bio-accumulative (B) are not required to submit special data. The properties of substances can be preliminarily screened by the following data:
- If the log Kow<4.5, the registered can be preliminarily considered as not bio-accumulative. If necessary, enterprises may determine the properties of substances based on the bio-accumulative test data;
- If the registered substances are readily bio-degradable, then the substances are not persistent (not applicable to inorganic substances). If the substances are not readily bio-degradable (except those used as pesticide, pharmaceutical or veterinary intermediates only), it is suggested that enterprises should carry out further degradation tests, such as intrinsic bio-degradation tests and pH related hydrolysis tests to judge the properties of substances;
3. Collect Information and Carry Out Risk Assessment
To successfully pass the environmental risk assessment, related enterprises must collect the full life circle information of the registered substances, including the manufacturing, processing, use and disposal information; scientifically evaluate the risk degree, formulate and implement the environmental risk control measures based on the the environmental and health hazard characteristics of new chemical substances and the actual exposure situation;
The preparation of environmental risk assessment report requires the physicochemical data, health toxicological and eco-toxicological data to evaluate the inherent hazards of substances. Besides, enterprises should also provide detailed information, such as the main reaction formula, technological process, raw and auxiliary materials and material balance, the generation of three wastes, the content of new substances and pollution control measures.
4. Optimize Model Parameter and Make Necessary Adjustments based on Actual Situations
When conducting environmental exposure assessment, exposure factors such as activity amount, emission coefficient, emission time and frequency of chemical substances should be set based on the actual situation. If necessary, targeted environmental risk control measures should be strengthened to eliminate unreasonable environmental risks. Chemical Exposure Assessment Tool (CET) is usually used to estimate local scale environmental and health exposure under various exposure scenarios. CET is a supporting technical tool of Technical Guidelines for Environmental and Health Exposure Assessment of Chemical Substances (Trial) The initial assessment shall be conducted by using the estimation method and recommended exposure parameters specified in the Guidelines (Trial). If the risk characterization value is greater than 1, some parameters such as emission time and emission mode can be adjusted appropriately based on the actual situation.

