
Polymers have been widely used in a wide range of industries such as packaging and coatings, construction materials, consumer goods, electrical and electronic industry, medical equipment and agriculture. For downstream users and formulators in the chemical industry, polymers are often used as surfactants, fluid modifiers, thickeners, emulsifiers, stimuli-responsive agents, conditioners, foam stabilizers, and so on.
The extensive number and the daily use of polymers make it more important for the governments and organizations to regulate them while not hindering innovation.
In Europe, polymers are mainly regulated by REACH & CLP.
Definition of Polymer
Europe has adopted the OECD definition of polymer. A polymer must meet the following three criteria:
- Molecules must be distributed over a range of molecular weights;;
- The weight percentage of molecules containing three monomer units or above should exceed 50%;
- The weight percentage of any molecule of the same molecular weight shall not exceed 50%;
The preferred method to determine whether a substance falls under the definition of a polymer is Gel Permeation Chromatography (GPC). Guidelines on the determination of the number average molecular weight (Mn) and molecular weight distribution using GPC are available in the OECD TG 118 (1996).
The best example available to explain the definition of a polymer can be taken from ECHA guidance document on polymers. This example is listed as below:
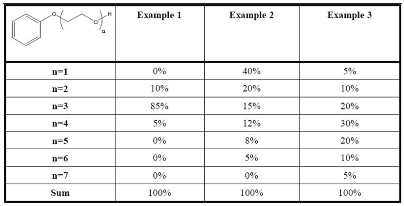
In Example 1, the substance consists of 10% ethoxylated phenol with n=2, 85% with n=3 and 5% with n=4. Since this substance comprises 85 weight percent of the same polymer molecule (n=3), it does not meet the definition of a polymer. Therefore, it should be considered as a standard substance.
In Example 2, only 15+12+8+5=40 weight percent of the substance consists of polymer molecules, i.e. molecules for which n>=3. For this reason, example 2 does not comply with the criteria for polymer definition either. Therefore it should also be considered as a standard substance.
Example 3, meets the definition of a polymer since 20+30+20+10+5=85 weight percent of the substance consists of polymer molecules (i.e. molecules for which n>=3) and none of the different constituents are present at concentrations above 50 weight percent (each constituent having a different molecular weight).
Polymer and REACH
EU does not differentiate between new polymers and existing polymers. All polymers are exempt from registration and evaluation under REACH. However, any manufacturer or importer of a polymer shall submit a registration to the European Chemicals Agency (ECHA) for the monomer substance (s) or any other substance (s), that have not already been registered by an actor up the supply chain, if both the following conditions are met:
- The polymer consists of 2 % weight by weight (w/w) or more of such monomer Substance (s) or other substance(s) in the form of monomeric units and chemically bound substance(s);
- The total quantity of such monomer substance(s) or other substance(s) makes up to 1 tonne or more per year.
Non-EU manufacturers of polymer may submit a registration to ECHA via a REACH only representative.
Please note that for a substance that does not meet the definition of polymer (for example, dimers, trimers), a registration dossier for the whole substance shall be submitted.
Any monomer is by definition an intermediate. Nonetheless, the specific provisions for the registration of intermediates under REACH do not apply to monomers. Only additives used to preserve the stability of the polymer and impurities are regarded as one part of the polymer and do not need to be registered separately under REACH. If other additives have been added to improve the performance of the polymer (for example, flame retardant), those additives shall be registered separately if the concentration of such additives are above 2% w/w and the annual quantity of such additives are above 1 ton per year.
Polymers may also be subject to authorization and restriction under REACH.
No-longer Polymers
Please note that the definition of a polymer in EU is different from the OECD definition before the 7th amendment of Directive 67/548/EEC was adopted in 1992. As a consequence of the implementation of this amendment, some substances which were considered to be polymers under the reporting rules for EINECS are no longer considered to be polymers under the 7th amendment. Therefore, these substances are called “No-Longer Polymers” (NLP). Those no-longer polymers shall be registered as a normal phase-in substance.
The NLP list mainly consists of the following groups:
- alkoxylated substances
- oligomeric reaction products
- oligomers from one monomer only
- dimers and trimers
- polymer-like substances containing 50% or more by weight of species with the same molecular weight
More information on NLP substances can be obtained through the website of the European Commission Joint Research Centre
Polymer and CLP
Polymers are not exempt from CLP regulation. A polymer is a substance and must be notified if it fulfills the criteria for classification as hazardous and it has been placed on the market. Importing a polymer does not correspond to the placing on the market of the monomers and any other substance from which the polymer substance originates. The C&L notification provisions for the import of a polymer can therefore only apply to the polymer substance itself.
Contact

