It can be hard to keep track of regulatory deadlines, so the CIRS team has put together this helpful infographic to make it a little bit easier. Scroll down to the bottom to find links to each of the dates mentioned.

April 12 – Canadian Requirements on Fragrance Allergen Disclosure Apply
- All cosmetic products (new and existing) must disclose 24 specific fragrance allergens when present above the threshold concentrations (Rinse-off products ≥ 0.01%; Leave-on products ≥ 0.001%)
May 1 – Updated CLP Classification Rules for Mixtures Apply (EU)
- Newly placed chemical mixtures must follow updated classification and labelling rules under amendments to the CLP Regulation.
- Impacts hazard communication, SDS updates, and classification reviews.
May 1 – Cosmetics CMR Restriction Update Takes Effect (EU)
- Amendments to the EU Cosmetics Regulation restricting certain carcinogenic, mutagenic and reprotoxic (CMR) substances become applicable.
May 1 – Cosmetic Products Containing 4-MBC Banned on EU Market.
· Sales ban for products containing the UV filter 4-Methylbenzylidene Camphor (4-MBC). Products must be removed from the EU market entirely.
May 1 – China’s Hazardous Chemicals Safety Law Takes Effect
- The regulation upgrades hazardous chemical safety management from administrative rules to formal national law. Full-process supervision, consolidated accountability, and enhanced safety obligations become mandatory.
May 19 – Extended Hazard Communication Compliance Deadline for Chemical Substances (U.S.)
- Newly manufactured chemical substances must update their SDS and labels to comply with the revised Hazard Communication Standard (HCS), aligned with GHS Rev. 7/8.
May 31 – Microplastics Reporting (EU)
- Manufacturers and industrial downstream users of synthetic polymer microplastics (SPM) (pellets, flakes, powders) used as feedstock in plastic manufacturing at industrial sites must submit an annual report on emissions to ECHA.
· Decree No. 280 of GACC takes effect, and Decree No. 248 of GACC is simultaneously repealed.
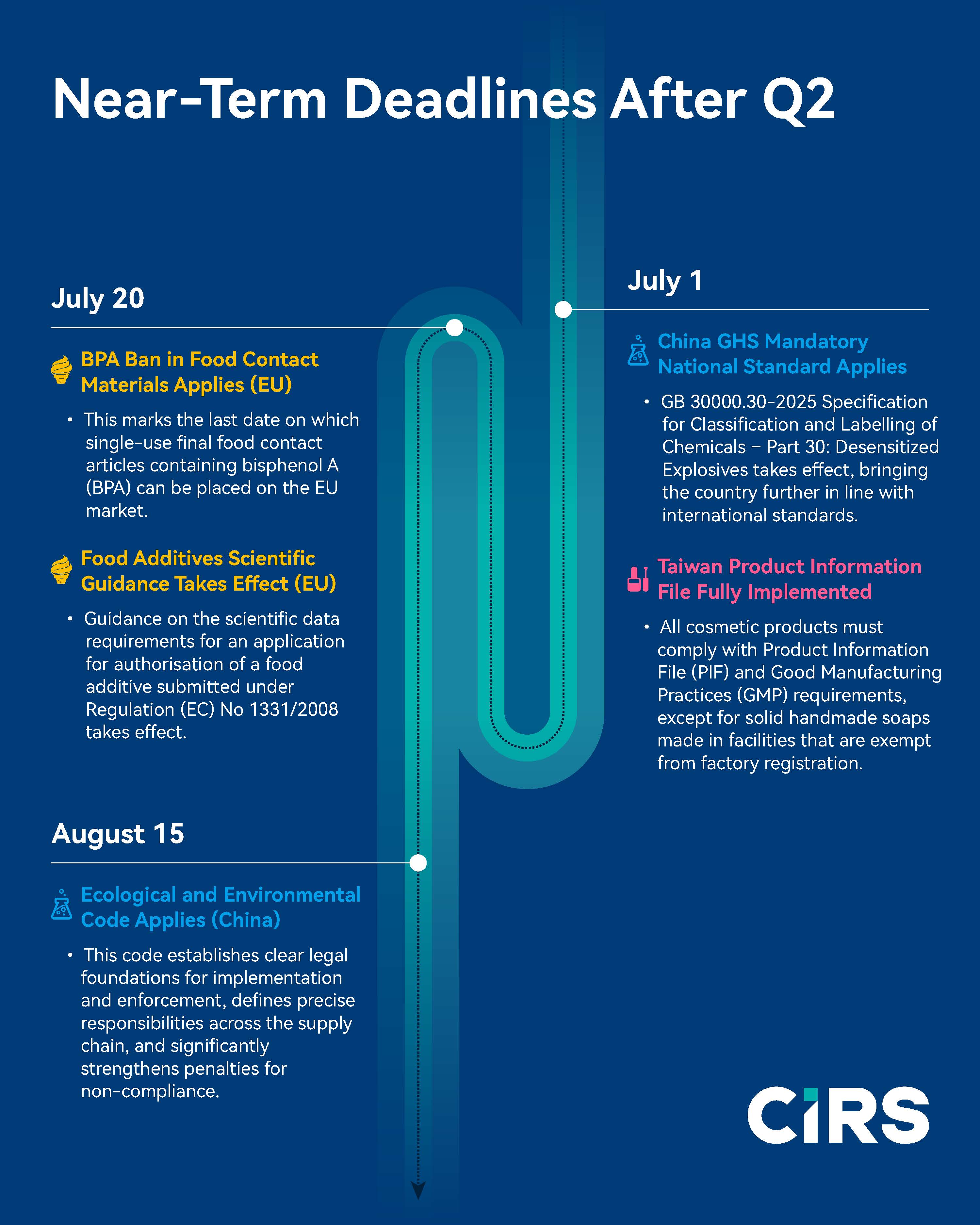
Near-Term Deadlines After Q2
July 1 – China GHS Mandatory National Standard Applies
- GB 30000.30-2025 Specification for Classification and Labelling of Chemicals – Part 30: Desensitized Explosives takes effect, bringing the country further in line with international standards.
July 1 – Taiwan Product Information File Fully Implemented
- All cosmetic products must comply with Product Information File (PIF) and Good Manufacturing Practices (GMP) requirements, except for solid handmade soaps made in facilities that are exempt from factory registration.
July 20 – BPA Ban in Food Contact Materials Applies (EU)
- This marks the last date on which single-use final food contact articles containing bisphenol A (BPA) can be placed on the EU market.
July 20 – Food Additives Scientific Guidance Takes Effect (EU)
- Guidance on the scientific data requirements for an application for authorisation of a food additive submitted under Regulation (EC) No 1331/2008 takes effect.
August 15 – Ecological and Environmental Code applies (China)
· This code establishes clear legal foundations for implementation and enforcement, defines precise responsibilities across the supply chain, and significantly strengthens penalties for non-compliance.

