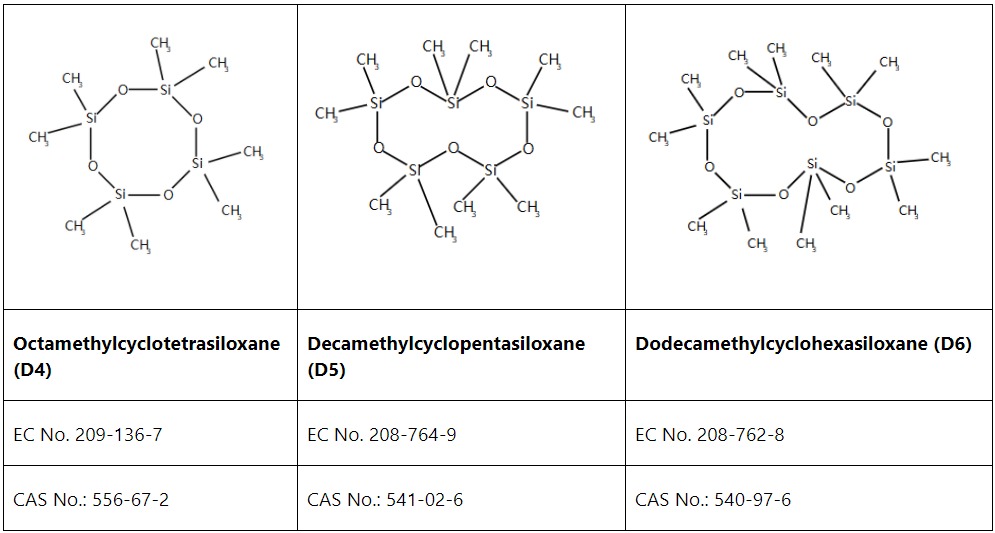
On November 27, 2025, the European Commission issued notification G/TBT/N/EU/1172 to the World Trade Organization (WTO) Technical Barriers to Trade Committee, announcing a planned technical update to the Classification, Labelling and Packaging Regulation (CLP, EC 1272/2008). The update involves addition of 38 new entries and revision to 10 existing entries in Annex VI.