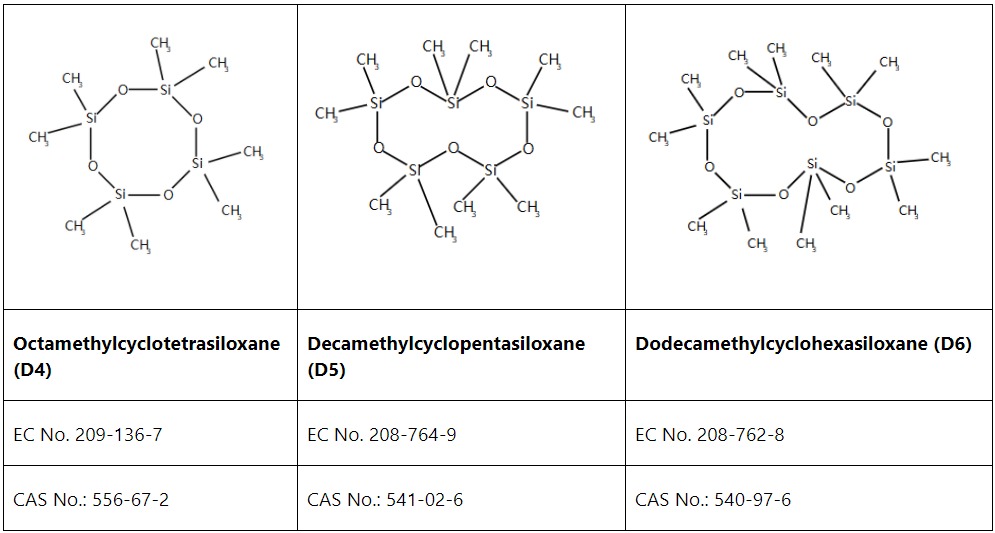
On March 11, 2026, the UK Health and Safety Executive (HSE) launched its first public consultation on adding Substances of Very High Concern (SVHCs) to the UK REACH Candidate List since Brexit. The consultation includes 15 proposed SVHCs, and companies have until April 20, 2026, to submit comments or supporting information. For companies placing chemicals on the market in Great Britain, this is a meaningful signal of how UK REACH is evolving.