Taiwan has initiated the annual reporting of new and existing substances since 1 Apr. 2021. Under the Provisions on the Registration of New and Existing Substances (2019), registrants must report the actual manufacturing/ import volume of last year of the new and existing substances between 1 April and 30 September annually since 1 April 2020. Information that shall be reported include: registrant information, the registration number, annual manufacturing/ import volume, etc.
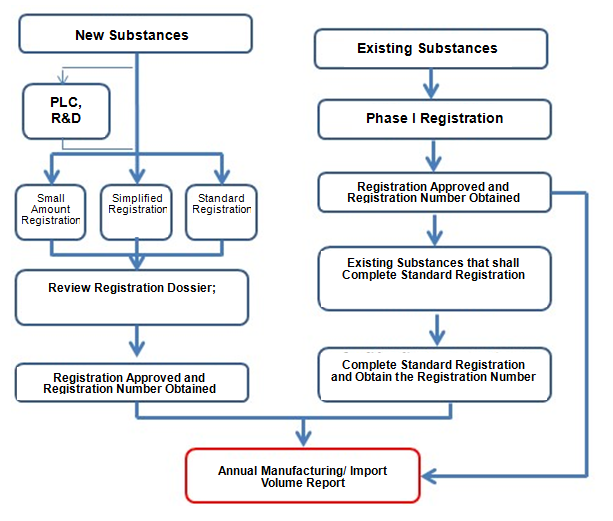
The Annual Report System covers two types: new substances and existing substances. The system will list the names of substances that shall complete report. Registrants can choose to complete report by themselves or authorize a representative to complete report.
Besides, as many registrants have met various problems during annual report for the Year 2019, the Report System has been updated.
The Report System is shown below:
The Annual Report System opens on 1 April each year and will be automatically closed on 1 October. Registrants need to complete annual volume report during this period. In case registrants fail to report the volume of the chemical substances or update the dossiers before 30 September, they shall submit application to the competent authority for permission to login the Report System and supplement the information within 30 working days.
If you have any needs or questions, please contact us at service@cirs-group.com.
