On July 27, 2023, the United Nations Economic Commission for Europe (UNECE) published the Globally Harmonized System of Classification and Labelling of Chemicals (GHS Rev.10). Revisions have been made to several parts, including:
- the classification procedure;
- the use of non-animal testing methods;
- precautionary statements; and
- annexes 9 and 10.
UN GHS refers to the Globally Harmonized System of Classification and Labelling of Chemicals (GHS), also referred to as the purple book. It is an international system created by the United Nations Committee of Experts on GHS, the International Labor Organization (ILO), and the Organization for Economic Co-operation and Development (OECD) to address the classification of chemicals by types of hazards and harmonize hazard communication elements, including labels and safety data sheets. It aims at providing a basis for the harmonization of rules and regulations on chemicals at national, regional, and global level, furthermore, trade facilitation is also an important factor. The UN GHS first edition was released in 2003. From then on, United Nations Committee of Experts on GHS meets regularly to discuss the update of UN GHS and releases a revised GHS edition every two years.
In comparison with GHS Rev.9, significant changes have been made in GHS Rev.10. For example, new provisions have been formulated.
In this article, we provide explanations from the following perspectives:
- physical hazards;
- health hazards;
- environmental hazards;
- hazards to the aquatic environment in Annexes; and
- hazard statements and precautionary statements.
I. Physical Hazards
1. Chapter 2.1, defines pyrotechnic substances or mixtures as explosive substances or mixtures. The explosive or pyrotechnic effect is also a new addition. It is an effect produced by self-sustaining exothermic chemical reactions including shock, fragmentation, projection, heat, light, sound, gas, and smoke.
2. Chapter 2.6, introduces the open-cup test method for flammable liquid. Open-cup tests are acceptable for liquids that cannot be tested in closed-cup test methods (due to their viscosity) or when open-cup test data is already available. In these cases, 5.6℃ should be subtracted from the measured value, as open-cup test methods generally result in higher values than closed-cup methods. However, in GHS Rev.9, closed-cup tests are only accepted in special circumstances.
3. In Chapter 2.7, metal powders refer to powders of metals or metal alloys.
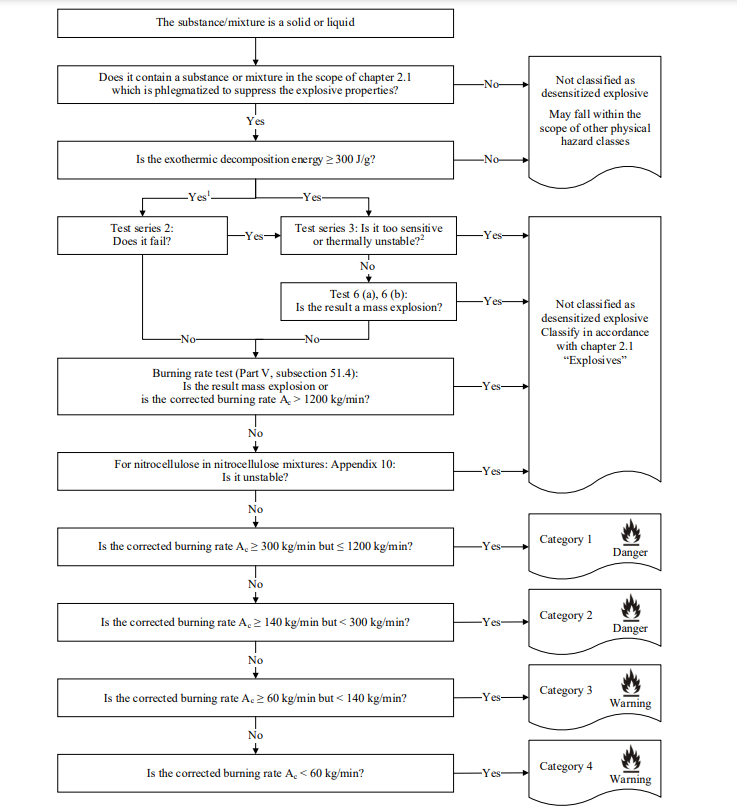
4. The definition of desensitized explosives is clearer than before. Desensitized explosives refer to substances and mixtures in the scope of chapter 2.1 which are phlegmatized to suppress their explosive properties in such a manner that they meet the criteria as specified in 2.17.2 and thus may be exempted from the hazard class “Explosives”. Moreover, “Test series 3: Is it too sensitive or thermally unstable?” is added to the decision logic of desensitized explosives. Pictograms and warnings for explosives in chapter 2.1 from GHS Rev.9 have been deleted.
II. Health Hazards
1. In Chapter 3.1 Acute toxicity, the formula is given to test inhalation toxicity under different exposure duration. The accepted exposure times for conversion range from 30 minutes to 8-hour exposures.

2. The use of non-animal testing methods for the classification of health hazards is clarified especially in Chapter 3.2 (Skin corrosion/irritation), Chapter 3.3 (Serious eye damage/eye irritation), and Chapter 3.4 (Respiratory or skin sensitization). Non-test methods include computer models predicting qualitative structure-activity relationships (structural alerts, SAR) or quantitative structure-activity relationships (QSARs) and read-across using analog and category approaches.
3. In Chapter 3.2 Skin corrosion/irritation, conclusions on no classification from read-across and QSAR shall be well substantiated. Conclusive non-test data for skin corrosion may be used for the classification of effects on the eye.
4. Chapter 3.4 Respiratory or skin sensitization, adds classification based on human data. A substance is classified as a skin sensitizer in category 1 if there is evidence in humans that the substance can lead to sensitization by skin contact in a substantial number of persons. In 3.4.2.2.3 Classification based on standard animal data, the classification of skin sensitization category 1 and sub-category can be made based on the results of radioisotopic local lymph code assay (LLNA).
III. Environmental Hazards
Certain words and sentences have been revised to make the description more accurate and explicit. For example, at the end of Chapter 4.1, “specific label elements for substances and mixtures classified into this hazard class based on the criteria in this chapter” is given and this sentence is also presented in most chapters in GHS Rev.10.
IV. Hazards to the aquatic environment in Annexes
Annex 9 adds guidance on the classification of organometallic compounds and organometallic salts. In order to classify a mixture containing Acute 1 and Chronic 1 ingredients, the classifier needs to be informed of the value of the M factor in order to apply the summation method.
V. Hazard statements and precautionary statements
1. It is permitted to combine more than one health hazard statement of equivalent severity if, for example, there is insufficient space on the label. For example, the hazard statement of H317+H340+H350 indicates “May cause an allergic skin reaction, genetic defects, and cancer”.
2. The hazard statement of H315+H319 indicates “causes skin irritation and serious eye irritation”.
3. Precautionary statements are revised or added in several hazard classifications under GHS Rev.10. For example, P262, P264, and P270 are added to acute dermal toxicity-category 3. Substantial changes are made to the prevention precautionary statements of respiratory sensitization highlighted in the following table:
Prevention | Response | Storage | Disposal | ||||
Rev. 9 | Rev. 10 | Rev. 9 | Rev. 10 | Rev. 9 | Rev. 10 | Rev. 9 | Rev. 10 |
P261 P284 | P233 P260 P271 P280 P284 | P304 + P340 P342 + P316 | P304 + P340 P342 + P316 | - | P403 | P501 | P501 |
This revised version significantly enhances the explicitness and clarity of the classification procedure, which holds great significance for the GHS classification of chemicals. GHS Rev. 10 also offers fresh insights for the formulation and updates of GHS regulations in different countries, providing a more scientifically grounded approach to preparing hazardous chemical labels and safety data sheets (SDS).
If you need any assistance or have any questions, please get in touch with us via service@cirs-group.com.
Further Information

