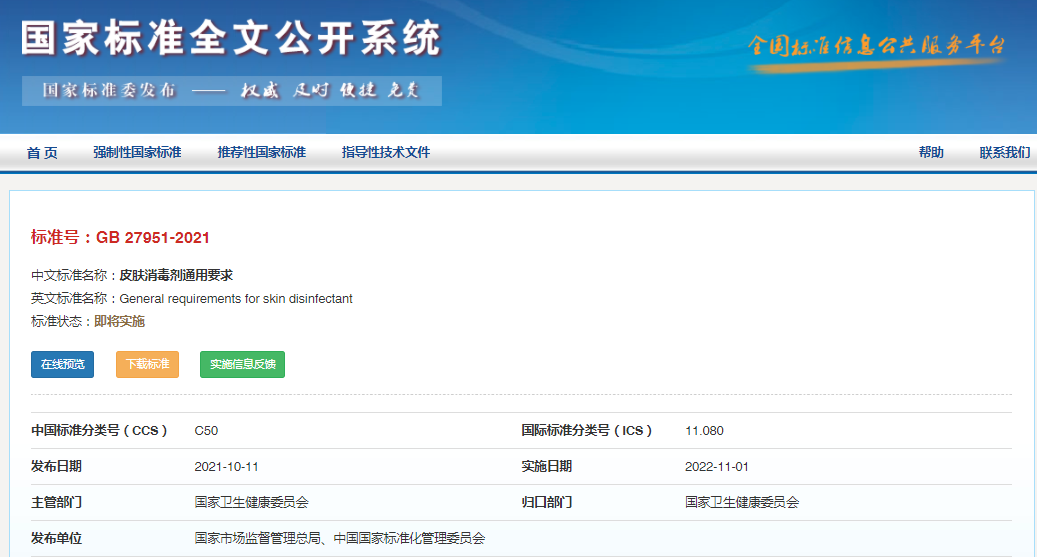
On October 11, 2021, the State Administration for Market Regulation (SAMR) and Standardization Administration (SAC) jointly published the mandatory standard – GB27951-2021 General Requirements for Skin Disinfectant. This standard will be implemented on November 1, 2022, replacing and renaming the current standard – GB 27951-2011 Hygiene Requirements for Skin Disinfectant.
Skin disinfectants are commonly used and are classified as category 1 disinfectants – those with the highest risks that are strictly managed including those that are used for medical devices, skin, and mucosa. The standard makes the filing of skin disinfectants clearer. CIRS Group has made a comparison between the new and existing standards.
In the new standard, SAC has:
- Added and removed some normative references;
- Changed the definition of intact skin;
- Added requirements for ingredients;
- Removed the common type of disinfectants for intact skin and damaged skin;
- Removed sensory indicators;
- Changed 'product quality requirements' to 'technical requirements';
- Added multiple skin irritation tests, and deleted the acute eye irritation test and skin allergy test;
- Added several determination requirements, including for antibiotics, antifungals, and antiviral drugs.;
Summary
In addition to the technical changes, there are also some detailed changes as follows:
- In section 4.1 Active ingredients, it is clearly listed that “guanidine, iodine, quaternary ammonium, peroxides, alcohols and phenol disinfectants used for skin disinfection shall meet the requirements of GB/T 26367, GB/T 26368, GB/T 26369, GB/T 26371, GB/T 26373 and GB/T 27947”;
- The new standard includes quantitative indicators for microbial killing. As products in different forms may choose different methods for the microbial killing tests, setting multiple quantitative indicators is conducive to the development and diversity of disinfection products;
- In the product identification section, information that needs to be marked on the label and instruction book is clearly listed. At the same time, it clearly indicated in this section that product labels must comply with GB 38598 General Requirements for Label and Instruction Book of Disinfection Products,
Text of GB 27951-2021 General Requirements for Skin Disinfectant (in Chinese):
https://openstd.samr.gov.cn/bzgk/gb/newGbInfo?hcno=02F68B0DAE0794E83699A396886938E3
Requirements of the GB 27951-2021 General Requirements for Skin Disinfectant are clearer than its predecessor. Enterprises may find the specific requirements more easily. CIRS Group suggests that related enterprises pay attention to the standard and actively comply with it.
If you have any needs or questions, please contact us at service@cirs-group.com.

