CIRS held a free webinar (download) on 17th and 18th Apr, 2018 for the Latest Update of Cosmetic Regulations in China since 2017. The presentation is divided into two parts. The first part mainly focuses on the data analysis of China cosmetic market from regulatory point of view. The second part lists the main regulations issued since 2017.
Data of registered or recorded cosmetics with CFDA or local FDAs
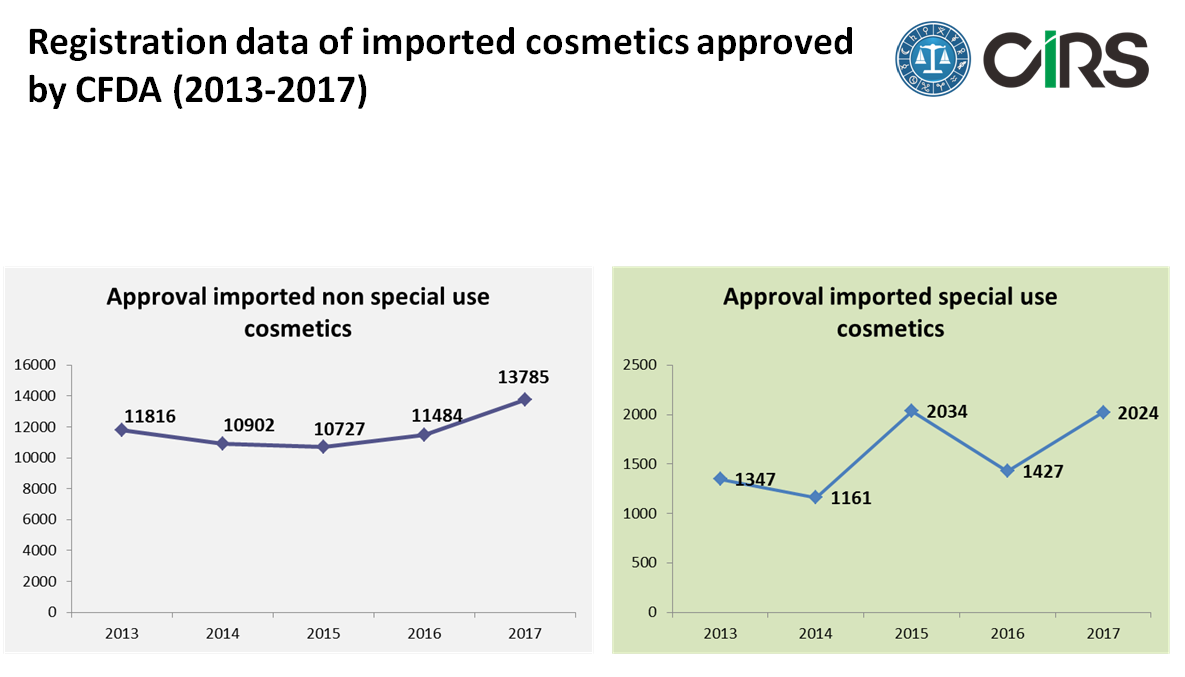
Import cosmetics by CFDA
For imported non special use cosmetics, the average number of approval imported non special use cosmetics is more than 11,000 from 2013 to 2017, . In 2017, the number of approval has a slight growth with reach to 13,785. Nevertheless, the number of approval imported special use cosmetics is about 15% of approval imported non special use cosmetics.
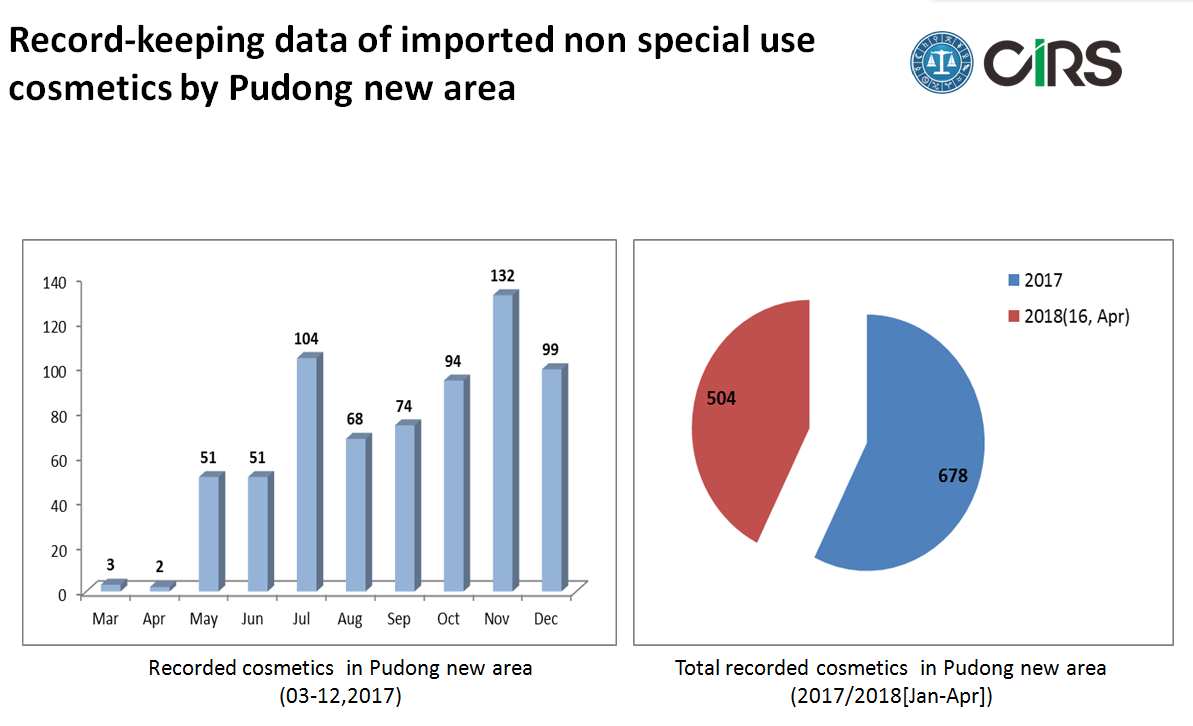
Import non special use cosmetics by Pudong new area
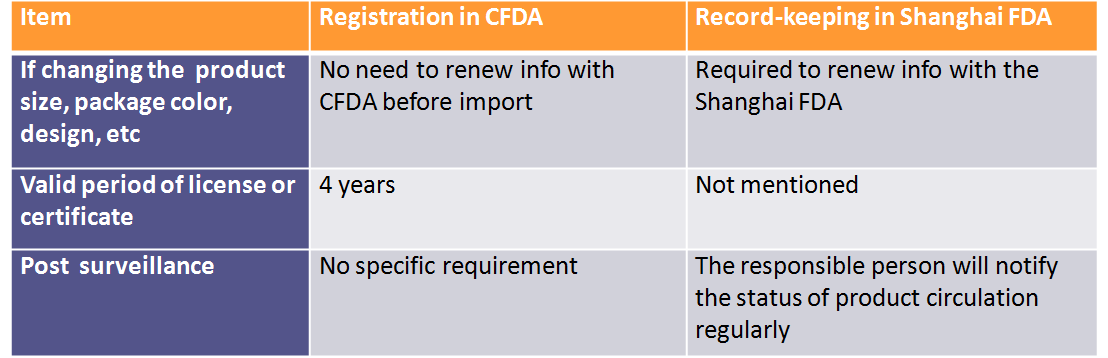
According to the pilot policy from 1st Mar, 2017 to 21st Dec, 2018, non-special use cosmetics, imported via Pudong new area, can be made record keeping with Shanghai FDA besides CFDA registration. So far, there are about 30% imported cosmetics recorded with Shanghai FDA.
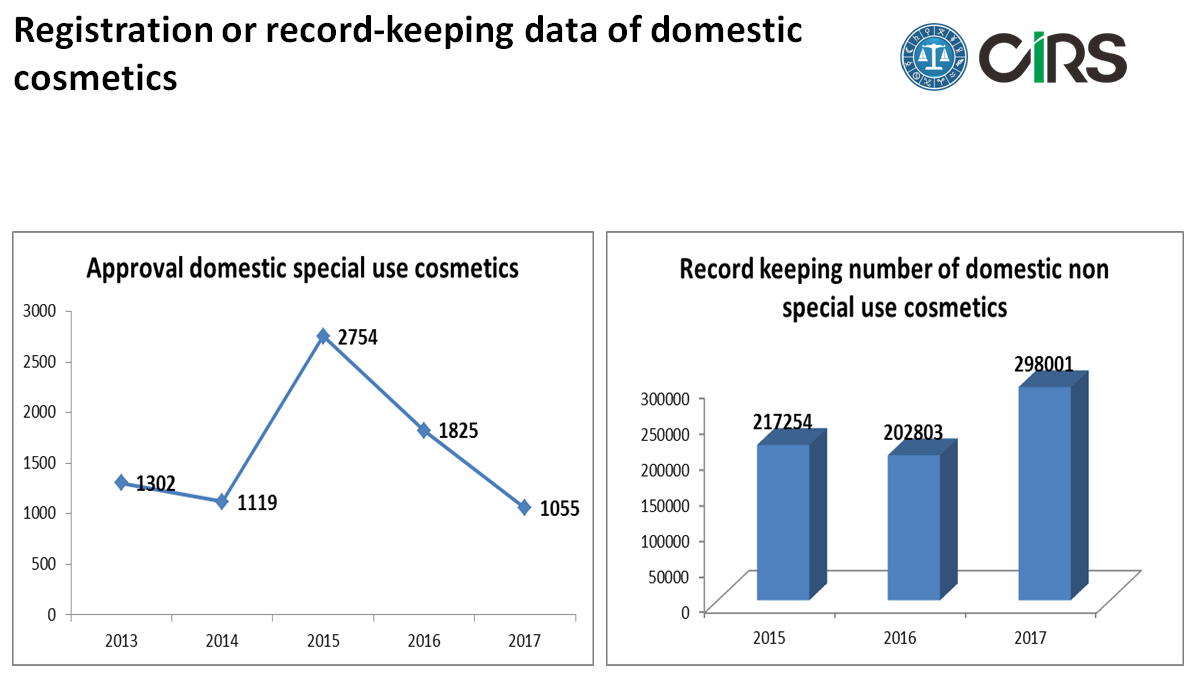
Domestic cosmetics by CFDA and local FDA
Compared with the imported special use cosmetics, the number of approval domestic special use cosmetics is similar. But there is a fast decline in 2016 and 2017. As we can see that the number of recorded domestic non special use cosmetics in 2017 will be closed to 300,000. There is no technical review process for the record keeping of local non special use cosmetics.
Updates of cosmetic regulations in China
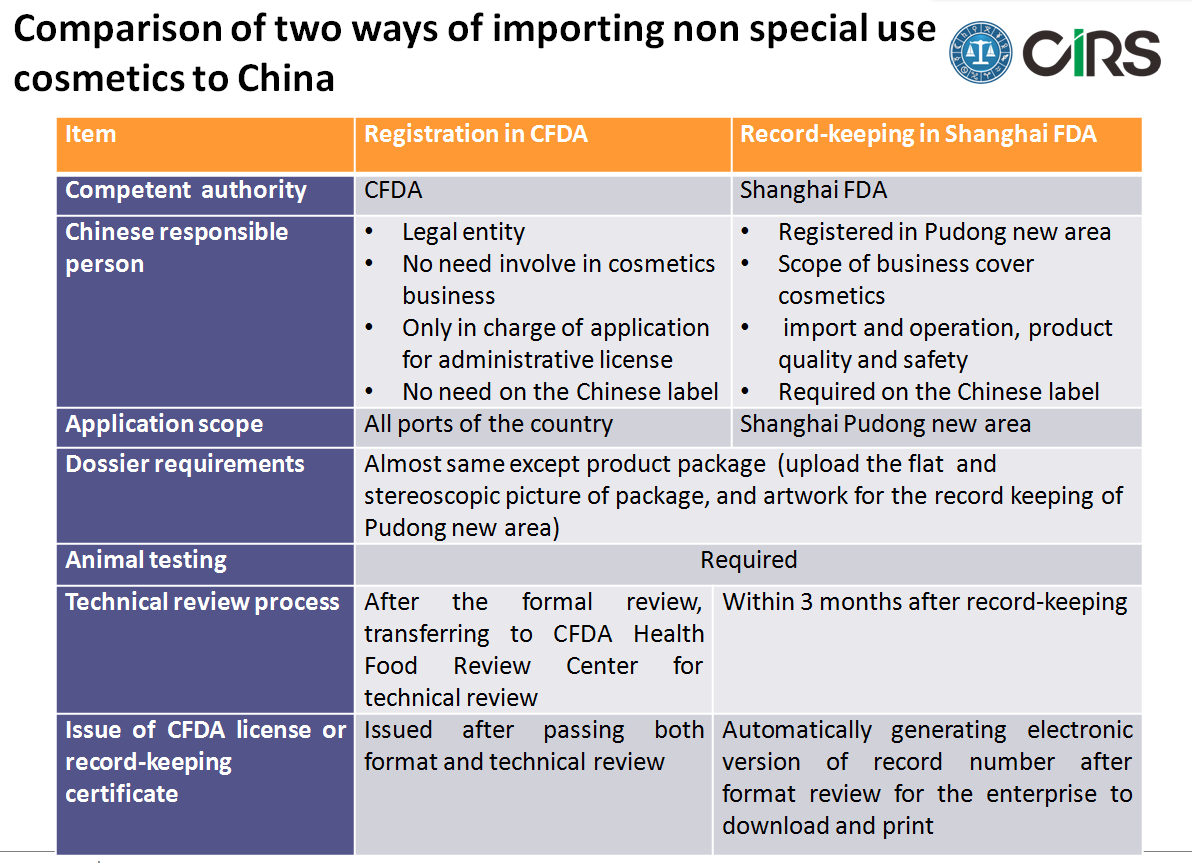
1) New record-keeping system for imported non special use cosmetics in Pudong new area
The presentation includes the background, related official files, conditions, general process and detailed comparison with the CFDA registration. The main differences can be found below.
2) Expanding 10 new free trade zones to implement new record keeping system of imported non special use cosmetics
This new pilot policy will be available from 8th Mar, 2018 to 21st Dec, 2018. The 10 new free trade zones include Tianjin, Liaoning, Zhejiang, Fujian, Henan, Hubei, Guangdong, Chongqing, Sichuan and Shanxi. Zhejiang and Guangdong FDA are the first two government bodies to issue notices for the preparation work of implementing this new pilot policy. Another presentation about this topic held by CIRS can be downloaded here.
3) New alternative methods available and consulted in public
On 21st Aug, 2017, there are two alternative methods accepted by CFDA. There are the In Vitro Skin Corrosion: Transcutaneous Electrical Resistance Test (TER) and Skin Photoallergy Test. Previously, only the In Vitro 3T3 Neutral Red Uptake Phototoxicity Assay (In Vitro 3T3 NRU PT) has been adopted on 11st Nov, 2016. In 2018, another two alternative methods In Chemico Skin Sensitisation: Direct Peptide Reactivity Assay (DPRA) and Short Time Exposure In Vitro Test (STE) are consulted in public on 5th Feb. As we can see, CFDA has sped up the process of adopting non animal test method in cosmetics and cosmetic ingredients.
4) Extending transition period of cross-border e-commerce
On 20th Sep 2017, Premier Li Keqiang highlighted in the State Council executive meeting the transition period of supervising import business of cross-border e-commerce will be extended to the end of 2018.
5) Imported cosmetics tariffs adjusted
On November 22, 2017, the Tariff Commission of State Council released a Notice regarding the adjustment of import tariff to some consumer goods. The adjusted import tariffs will come into force on December 1, 2017. The imported cosmetics tax rate is dropped from the original 15%, 10%, 9%, 6.5% to 5% and 2%.
6) Public opinion on new guidelines for cosmetic classifications
According to the new guidelines, the classification code will consist of 4 layers. They are the efficacy claim, application site, product form and suitable users. There will be 26 codes of efficacy claim, 27 of application site, 18 of product form, and 7 of suitable user. The detailed info can be found in our analytical article.

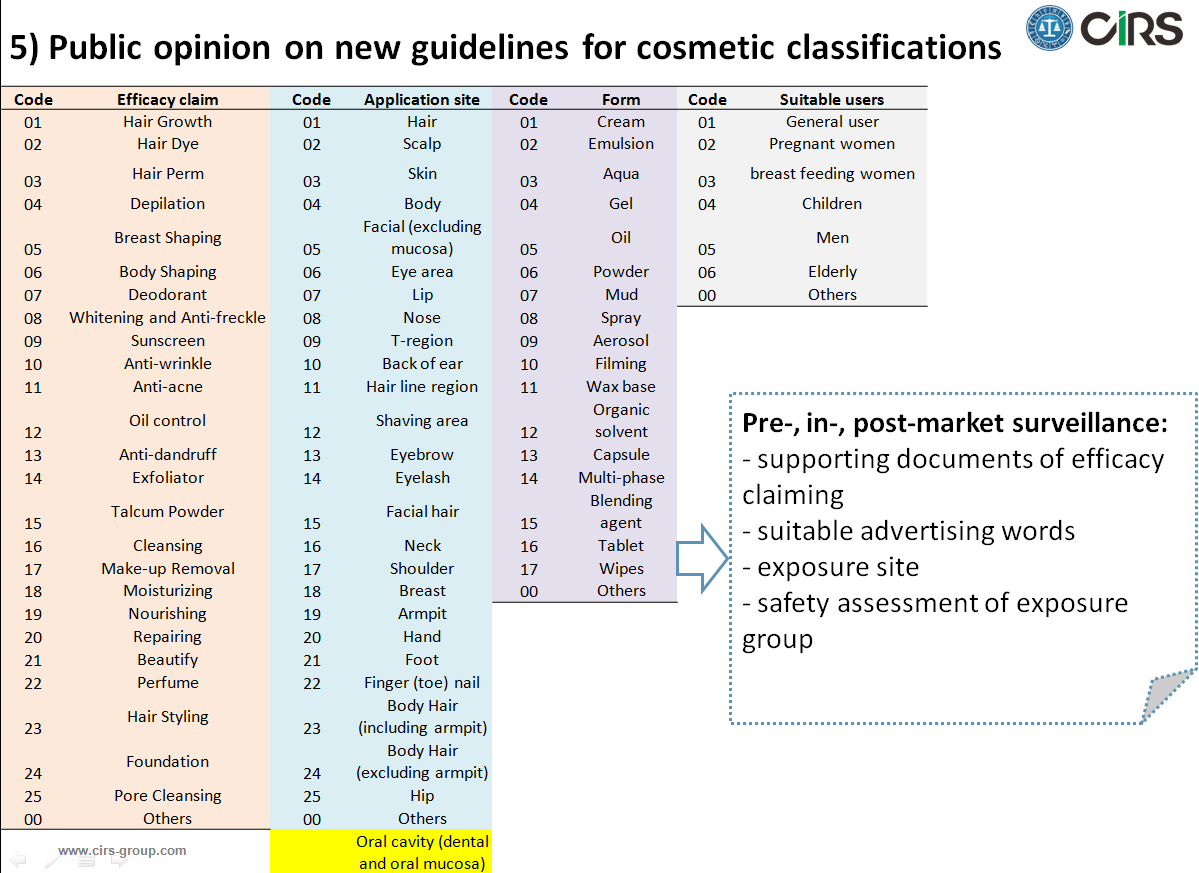
7) Guidelines for the evaluation of cosmetic claims (draft)
On Jan 24, 2018, the National Institutes for Food and Drug Control published the Guidelines for the Evaluation of Cosmetic Claims for public opinion. It is required to evaluate the specific function like sunscreen, anti-freckle, hair growth, breast shaping, fitness, deodorant, anti-aging, anti-acne, oil control, anti-dandruff, repairing, moisturizing (>2hs). The manufacturer can make the self-evaluation or appoint the third party lab to perform the evaluation. No matter whether the in vivo method on human/animal, or in vitro method, self-development method by verified lab would be acceptable.
If you have any needs or questions, please contact us at service@cirs-group.com.

