On May 31, 2021, NMPA issued the notice on Measures for Administration of Cosmetics Labeling (No.77, 2021), encouraging cosmetics registrants and filers to label cosmetics in accordance with the provisions of the Measures from the date of this announcement. From May 1, 2022, cosmetics applying for registration or filing must meet the provisions and requirements of the Measures; Where cosmetics that have previously applied for registration or been filed fail to be labeled and marked in accordance with the Measures, the cosmetics registrants and filers must complete the updates to product labels before May 1, 2023 to make them conform to the provisions and requirements of the Measures.
Definition
Cosmetics labels refer to words, symbols, numbers, patterns and other marks used to identify and explain the basic information, attributes and safety precautions of products on product sales packages, as well as packaging containers, packing boxes and instructions attached with identifying information.
Cosmetics registrants and filers are responsible for the legality, authenticity, integrity, accuracy and consistency of cosmetics labels.
Notes: In addition to text content, symbols, numbers, patterns and other marks in labels also belong to the contents of cosmetics labels. If other languages or symbols are used, corresponding explanations in standard Chinese characters shall be printed on the visible surface of product sales packaging.
Contents that Must Be Labeled
Product name in Chinese and registration certificate number of special cosmetics
Labeling requirements: The labels shall be printed in a prominent position on the visible surface of the package for sale, with at least one leading phrase leading to the Chinese name of the product.
Chinese names shall not use letters, Hanyu Pinyin, numbers or symbols, except for registered trademarks, sun protection index, color number or series number, or others that must be named with letters, Hanyu Pinyin, numbers or symbols. Where a registered trademark uses letters, Chinese pinyin, numbers, symbols, etc., its meaning shall be printed visually on the sales package of the product.
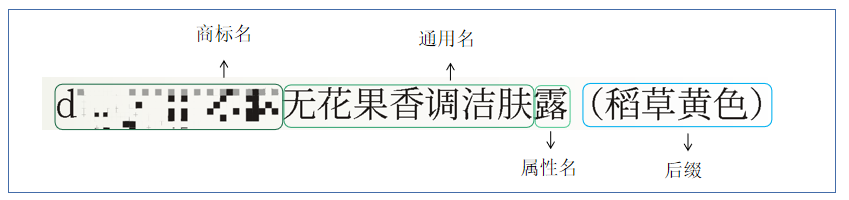
The Chinese name of cosmetics generally consists of three parts: brand name, generic name and attribute name.
Brand Name
- Claims on medical effect or the efficacy that the product does not have shall not be used as the Brand Name of the product;
- If expressions that imply the use of certain raw materials are used as the Brand Name of a product:
- The sales packaging of the product shall show the purpose of use in the visible surface, when the product contains such raw material;
- It shall be clearly marked on the visible side of the sales packaging that the product does not contain such raw materials, and relevant expressions shall only be used as the Brand Name when the product does not contain such raw materials.
Generic Name
- It is generally used to indicate the raw materials of the products or to describe the uses and used parts of the products.
- The specific names of raw materials or words indicating the category of raw materials can be used as generic name when the raw material of the products are consistent with the ingredients in the product formula, and the efficacy of the raw materials in the product are consistent with the efficacy claims of the product. For example, hyaluronic acid moisturizing cream, the function of hyaluronic acid in the product should be moisturizing, and the product should claim moisturizing effect. The two are consistent.
- Where the name of animal, plant or mineral is used to describe the flavor, color or shape of the product, the general name may use the name of animal, plant or mineral together with the flavor, color or shape, or the flavor, color or shape of products may also be indicated following the attribute name, such as rose aroma, strawberry color, etc., no matter the formula of products contain such raw materials or not.
Trademark Name
Generally indicate the true physical nature or form of the product.
Suffix
If the brand name, general name and attribute name of different products are the same, other information that need to be marked, including the color or color number, SPF, smell, applicable hair quality, skin quality or specific groups of people, etc., can be indicated following the attribute name.
If the brand name, general name or attribute name meets the above requirements when used alone, and may create ambiguity on the efficacy of the product when combined used, explanations should be given on the visible surface of the sales package.
The registration certificate number of special cosmetics refers to the registration certificate number issued by NMPA, and shall be marked on the visible side of the sales package.
If the registrant or the filer is an overseas enterprise, name and address of the Chinese responsible unit shall also be labeled;
According to the Cosmetics Supervision and Administration Regulations, cosmetics registrants and filers are responsible for the quality, safety and efficacy claims of cosmetics. Information of the registrant, filer and the Chinese responsible person shall cover the name and address that are specified in the product registration certificate or the filing information, and shall be led out with the corresponding leading words respectively.
The name and address of the manufacturer and the production license number of the manufacturer of domestic cosmetics;
The name and address of the manufacturing enterprise that is the last to touch the contents shall be provided. Where a registrant or a filer entrusts multiple production enterprises to complete the last step, the name and address of all entrusted production enterprise may be marked respectively, and the specific production enterprise of the product may be indicated by code or other means.
Standard serial number of product execution;
Mark the standard number of the product execution on the visible surface of the sales package, and lead out with the corresponding guide words.
Full composition
- The Standard Chinese name of the cosmetic ingredients shall be labeled on the visible surface of the sales package, with "ingredients" as the guide word. Ingredients shall be listed in descending order of their contents. All the ingredients not more than 0.1% (w/w) should be introduced as "other trace ingredients", and may not be listed in descending order of contents. (Other trace ingredients include ingredients with a content equal to 0.1% (w/w).
- Where a formula is made up of compounded or mixed raw materials, the content of each component in the formula shall be used as the basis for ordering and will be used to judge whether they are trace ingredients.

Net Content
The net content of cosmetics shall be calculated based on national legal units of measurement and marked on the display surface of the sales package.
Notes: According to GB 5296.3-2008 Instruction for Use of Consumer Products-General Labeling for Cosmetics, cosmetics in quantitative packaging mark the net content in accordance with AQSIQ Order 75. Since the regulations have not been abolished for the time being, the net content should be labeled in accordance with relevant requirements for the time being, and the font size of net content labeling should be implemented according to relevant requirements.
Use period
Labeling requirements: labeling in one of the following ways on the visible surface of the sales package with the corresponding guide words:
- Production date and shelf life: The production date shall be marked in Chinese characters or Arabic numerals in order of four-digit year, two-digit month and two-digit date, such as 20220101;
- Production lot number and expiration date.
For products with packaging boxes, in addition to the above methods, the production batch number and the usage period after opening can be marked on the packaging that directly contact the contents.
When the sales package contains multiple independent packaging products inside, each independent packaging shall be marked with the expiration date. The expiration date printed on the sales package shall be the earliest one among all the independently packaged products inside the sales package. It is also available to mark the service life of each independently packaged product separately.
Method of use
Labeling requirements: Label on the visible surface of the sales packages or attached to the product manual.
Other contents that shall be marked as required by laws, administrative regulations and compulsory national standards.
Under any of the following circumstances, "Notice" or "Warning" shall be used as the guiding language and warning words shall be marked on the visible surface of the sales package:
- Laws, administrative regulations, departmental rules, mandatory national standards and technical specifications regulate on the labeling of warning words and safety-related matters for limited or permitted components of cosmetics; For example: According to the Safety and Technical Standards for Cosmetics (2015), if salicylic acid is used in the formula, and the product is likely to be used by children under three years old and is in long-term contact with skin, it should be marked "containing salicylic acid; Not for children under three years of age”.
- Laws, administrative regulations, departmental rules, mandatory national standards and technical specifications regulate on the labeling of matters need attention for cosmetics applicable to children and other special groups. Provisions on the Cosmetics Supervision and Administration Regulations of Children requires that children's cosmetics should be marked with a “Children’s Cosmetic Mark” on the display surface of sales packaging as stipulated by the State Medical Products Administration. Children's cosmetics should be guided by "Notice" or "Warning", and warning words, such as "should be used under adult supervision", should be marked on the visible surface of sales packaging.
- Other warning words and precautions stipulated by laws, administrative regulations, departmental rules, mandatory national standards and technical specifications. For example: According to Cosmetic Safety Technical Specification (2015), benzoic acid and its sodium salt and other raw materials can be used as preservatives. However, when these substances are used for other purposes, the raw materials and their functions should be marked on the label. Furthermore, in terms of perfume products or aerosol can products containing a large amount of ethanol, it is suggested to judge whether they are flammable or explosive based on the "Specification for The Classification and Labeling of Chemicals".
Prohibited Content
(1) Medical terms, names of medical celebrities, words describing medical functions and effects, names of approved drugs to express or imply that the product has medical effects;
(2) False, exaggerated or absolute terms to describe falsely or misleadingly;
(3) Alluding to medical effects or making false claims by means of trademark, pattern, font color size, color difference, homophony or suggestive words, letters, Hanyu Pinyin, numbers or symbols;
(4) Fabricating concepts with terms and mechanisms that have not been widely accepted by the scientific community to mislead consumers;
(5) Misleading consumers by fabricating false information or belittling other legitimate products;
(6) Misleading consumers by using fictional, forged or unverifiable scientific research achievements, statistical data, survey results, abstracts, quotations and other information;
(7) Implying functions of the product that the product does not have or is not allowed to claim by claiming the functions of the raw materials used in the product;
(8) Using marks and awards that have not been confirmed by the competent authorities of the relevant industry to make claims and expressions related to the safety and efficacy of cosmetics;
(9) Using the names and images of state organs, public institutions, medical institutions, public welfare institutions, their staff members and appointed experts as certificates or recommendations;
(10) Assertions or guarantees on efficacy or safety of products;
(11) Vulgar, feudal superstition or other contents that violate public order and good customs;
(12) Other contents prohibited by laws, administrative regulations and compulsory national standards for cosmetics.
Other labeling requirements specified by the cosmetic label
(1) The minimum sales unit of cosmetics shall contain labels.
The labels shall comply with the requirements of relevant laws, administrative regulations, departmental rules, compulsory national standards and technical specifications, and the contents shall be legal, true, complete and accurate, and shall be consistent with the relevant contents of product registration or filing. When the product is packaged in small packs with the net content not more than 15 g or 15 ml, information such as the Chinese name, serial number of registration certificate for special cosmetics, as well as name, net content, use period, etc. of the registrant or filer are required to be labeled on the visible surface of the sales packages; Other information required can be marked in the specification attached to the products. These Measures shall apply to the labeling of cosmetics provided to consumers in the form of free trial, gift or exchange. That is, the labels on the sales packages of cosmetics used for non-sales purposes shall conform to the labeling requirements under the Measures for the Administration of Cosmetics Labels.
(2) Use standard Chinese characters in cosmetic labels.
If other characters or symbols are used, corresponding explanations in standard Chinese characters shall be printed on the visible surface of the product sales package, except for websites, names and addresses of overseas enterprises and commonly accepted professional terms that must be used in other characters. The font size of words in other languages on the identical surface with the Chinese label shall be smaller than or equal to that of the corresponding standard Chinese characters (trademarks excepted).
(3) If the cosmetic products paste Chinese labels (such as imported cosmetics put in China market), information on product safety and efficacy claims recorded in the Chinese labels shall be consistent with that of the original labels. That is, the claims about safety and efficacy of products sold in China are consistent with those in the country of origin.
(4) For products with packing boxes, Chinese name and the expiry date of the products shall also be marked on the packing containers that have direct contact with the contents.
Legal liability
Measures for Administration of Cosmetics Labeling
If the cosmetics labels make the following mistakes, but they do not affect the quality and safety of products and will not mislead consumers, the drug supervision and administration departments shall process in accordance with the requirements of Article 61 (2) of the Cosmetics Supervision and Administration Regulations:
- The font size of characters, symbols or numbers is not standard, there are some errors or omission in the Chinese characters or there are some non-standard Chinese characters;
- The format of the usage period and net content are not standardized;
- Cosmetics labels are not clear and difficult to recognize or read, or part of the printed words fall off or are not firmly pasted;
- The names of cosmetic ingredients are not standardized or the ingredients are not listed in descending order of content;
- Failing to use guide language in accordance with the provisions of the Measures;
- The Chinese name of the product is not marked in a prominent position;
- Other circumstances that violate the Measures but do not affect product quality and safety and will not mislead consumers.
Related enterprises shall be punished according to law, if labels of their products violate the Measures and meet the situations prescribed in Article 61 (5) of Cosmetics Supervision and Administration Regulations.
Cosmetics Supervision and Administration Regulations
Article 61 (2): Where there are defects in the labels of cosmetics produced or marketed but do not affect quality and safety and do not lead consumers astray, the regulatory authority shall order correction; Those who refuse to make corrections shall be fined not more than 2,000 yuan.
Paragraph 1 of Article 61 (5): Cosmetics whose labels do not conform to the provisions of these Regulations shall be confiscated by the department responsible for drug supervision and administration. Incomes arising from the sales of the above cosmetics, as well as the raw materials, packaging materials, tools, equipment and other articles specially used for illegal production and marketing shall also be confiscated; Where the value of cosmetics illegally produced or operated is less than 10,000 yuan, a fine of more than 10,000 yuan but less than 30,000 yuan shall be imposed; If the value of goods exceeds 10,000 yuan, a fine of not less than 3 times but not more than 10 times of the value of goods shall be imposed; If the circumstances are serious, related enterprises shall have to to suspend production or business and the filing or registration certificate will be revoked; the legal representative or principal person in charge of the units, the direct responsible person and other personnel with direct responsibilities shall fined more than 1 times and less than 2 times of the income obtained from the unit in the previous year, and prohibited from engaging in cosmetics production and operation activities for 5 years.
Case Study

If you have any needs or questions, please contact us at service@cirs-group.com.

