According to the Federal Food, Drug, and Cosmetic Act (FD&C Act), all color additives must obtain FDA approval before they can be used in food, drugs, cosmetics, and medical devices intended for sale in the United States. To sell new color additives or use approved color additives for new purposes, manufacturers must first submit an application to the FDA.
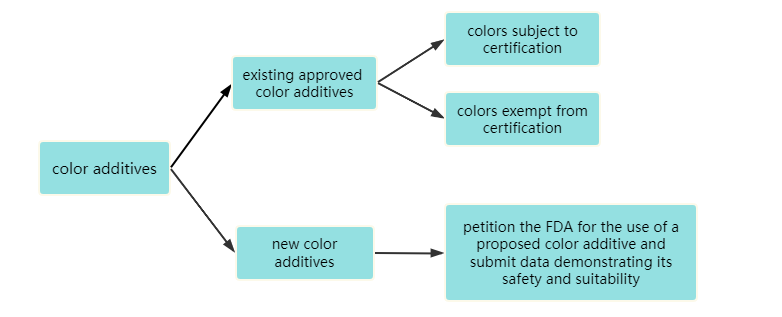
The FD&C Act Section 721(c) and color additive regulations separate approved color additives into two main categories: those subject to certification (sometimes called "certifiable") and those exempt from certification.
Colors subject to certification - These color additives are derived primarily from petroleum and are sometimes known as "coal-tar dyes" or "synthetic-organic" colors. Color additives must be batch certified by FDA if they are to be used in cosmetics (or any other FDA-regulated product) marketed in the U.S. Color additives must pass analysis of its composition and purity in FDA's own labs. For color additives that have passed batch certification but have been repackaged, repackaging batch certification is also required.
Colors exempt from certification - These color additives are obtained primarily from mineral, plant, or animal sources. They are not subject to batch certification requirements. However, they still are considered artificial colors, and when used in cosmetics or other FDA-regulated products, they must comply with the identity, specifications, uses, restrictions, and labeling requirements stated in the regulations.
Batch certification
When applying for batch certification, manufacturers are required to submit color additive samples and corresponding documents to the FDA. All foreign companies in the United States must designate a US agent in the batch certification application.
FDA Color Additive Batch Certification Process

Our Service
- FDA Color Additive Batch Certification; and
- US Agency Service.