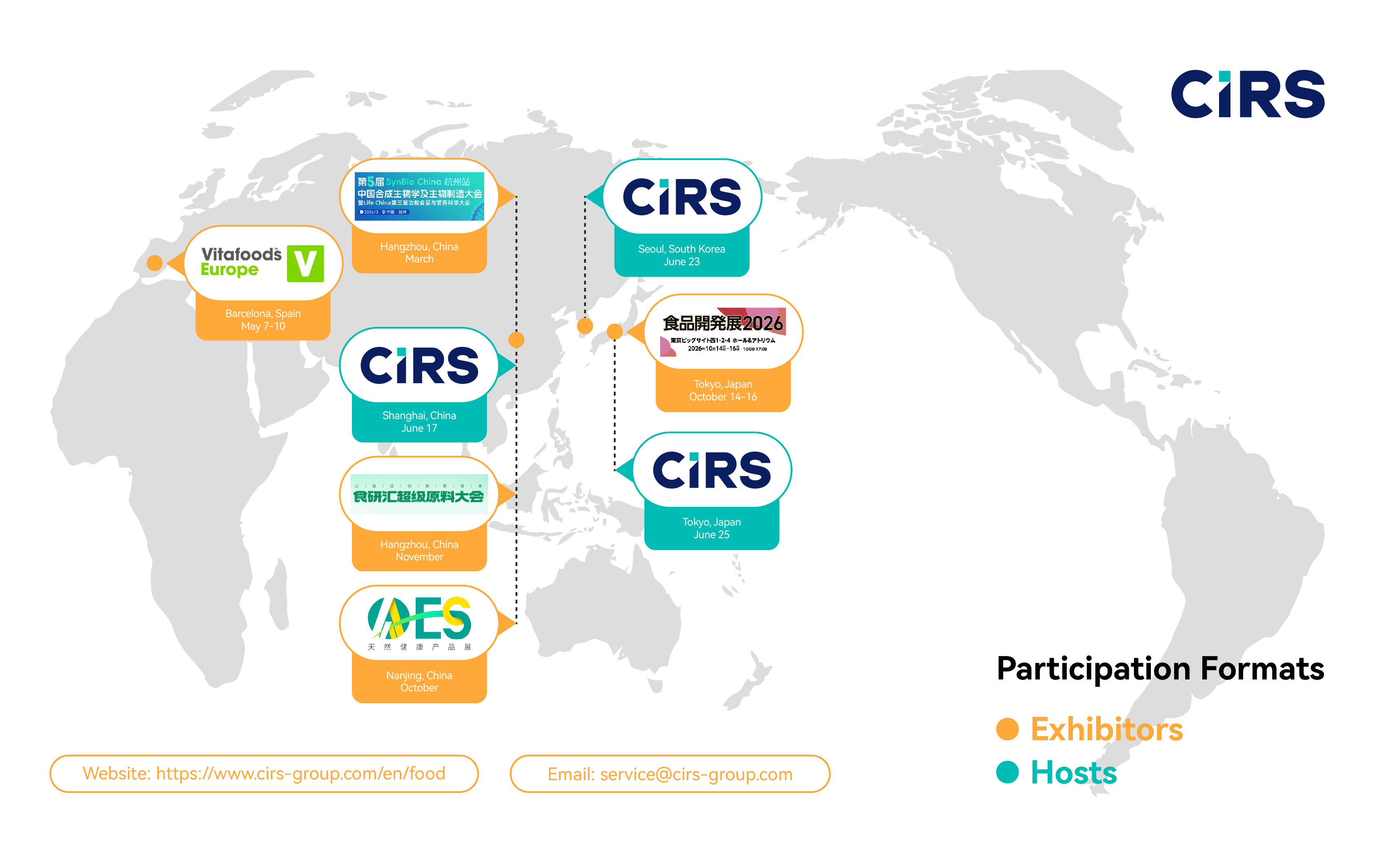
Meet our global food regulatory team at leading 2026 industry events worldwide. Explore our events calendar and book a meeting.



Meet our global food regulatory team at leading 2026 industry events worldwide. Explore our events calendar and book a meeting.

CIRS EU, the European subsidiary of CIRS Group, will be exhibiting at Vitafoods Europe in Barcelona, Spain, from May 5–7, 2026 (Booth No.: 6D84M, Hall 6) . CIRS has participated in Vitafoods Europe multiple times in Geneva, and attended the 2025 event in Barcelona. This year, we will once again be present at Vitafoods Europe 2026. We sincerely invite both new and existing partners to join us for discussions and to explore collaboration opportunities together. We look forward to seeing you there.

At the request of the European Commission, the Panel on Nutrition, Novel Foods, and Food Allergens (NDA) of the European Food Safety Authority (EFSA) was tasked with providing an opinion on the use of carbon dioxide extract from Cannabis sativa L. as a novel food under Regulation (EU) 2015/2283.

CIRS Group has compiled and summarized the intended uses and use levels of HMOs approved in the EU for industry reference. Among them, chemically synthesized and microbially produced 2’-FL have identical scope of use and use levels; similarly, chemically synthesized and microbially produced LNnT share the same conditions of use. The remaining HMOs are produced via microbial sources, and their intended uses may vary depending on the production organism.

In January 2026, the European Food Safety Authority released updated scientific guidance and administrative guidance for food additive authorisation applications. These updates reflect recent technological advancements in food additives. Compared with previous versions, the new guidance improves both comprehensiveness and clarity, offering more specific and actionable instructions.

Pasteurised Akkermansia muciniphila, also known as inactivated AKK, is a heat-treated, non-viable beneficial gut bacterium associated with metabolic improvement and enhanced intestinal barrier function. As a representative “postbiotic”, it has attracted significant attention.

On February 9, 2026, the European Food Safety Authority (EFSA) updated its safety statement regarding cannabidiol (CBD) as a novel food. The update establishes a provisional safe intake level for CBD with a defined scope of application, outlines data gaps in its safety research, and confirms that EFSA will continue to advance the risk assessment of CBD.

On February 3, 2026, the European Commission published a revision to the regulation governing the use of bisphenol A (BPA) in food contact materials and articles. The revised regulation entered into force on February 23, 2026. The revision aims to amend and clarify provisions in the earlier BPA regulation to address inconsistencies and improve regulatory clarity. It refines relevant definitions, clarifies transitional arrangements, and updates compliance requirements for the use of BPA in food contact materials and articles placed on the EU market, except for specific derogated applications.

Based on publicly available information from the Open EFSA and regulations issued by the European Commission, CIRS conducted a statistical analysis of the EU Novel Food (NF) approval status in 2025 for enterprise reference.
Based on publicly available information from the Open EFSA and regulations issued by the European Commission, CIRS conducted a statistical analysis of the 2025 EU food additive submissions for corporate reference.