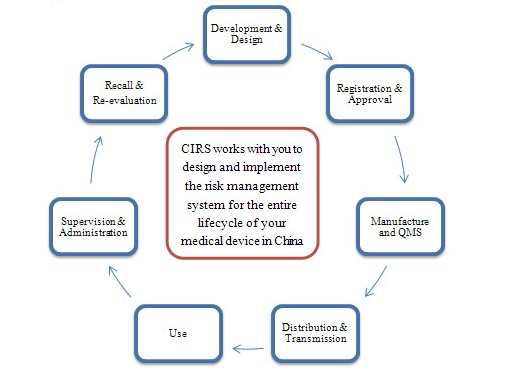
In medical device industry, the risk management is vital part of all your company’s processes, it involve in the entire lifecycle of a device. There are different risks raised from each process of a device marketed in China: market risk, regulatory risk and product quality risk. To ensure your company gets a safe and effective product to entry into Chinese market on time and reduce the regulatory risk, a successes implementation of risk assessment and management would be a best support for your business in China.
Risk Management Scheme
Please join in our “Perennial Regulatory Consultant Program”. For complex and urgent projects, we will assign a dedicated project manager to customize a program specific to your requirements. We are ready to be of service at any time.