On 5 September 2017, Former CFDA published the Dossier Requirements of Foods for Special Medical Purpose (Trial) (2017 revision), which specified the format and contents requirements of FSMP application dossiers. In order to help applicant have a better understanding on the dossier requirement, CIRS analyzes the application dossier requirements and makes a conclusion as following:
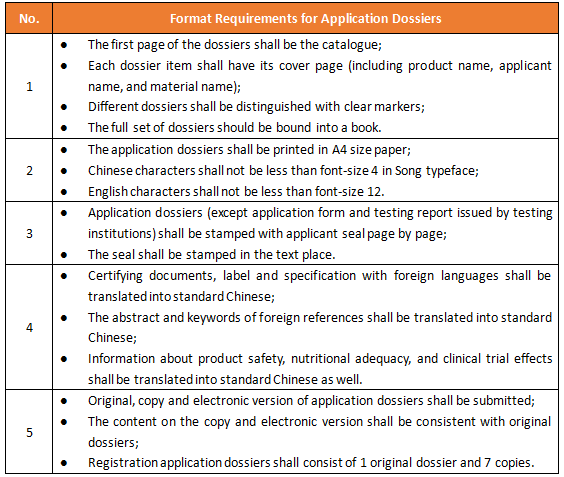
1. Format Requirements for Application Dossiers
2. Requirements for Each Item of Application Dossiers
CFDA Health Food Review Center (hereinafter called Review Center) will carry out the technical review of FSMP registration application dossiers. If the dossiers are unqualified, the Review Center will require applicant to supplement dossiers, or reject the application directly.
2.1 Application Form
Fill in the registration form through the FSMP registration system (http://ty.zybh.gov.cn/stzcww/) and print the application form.
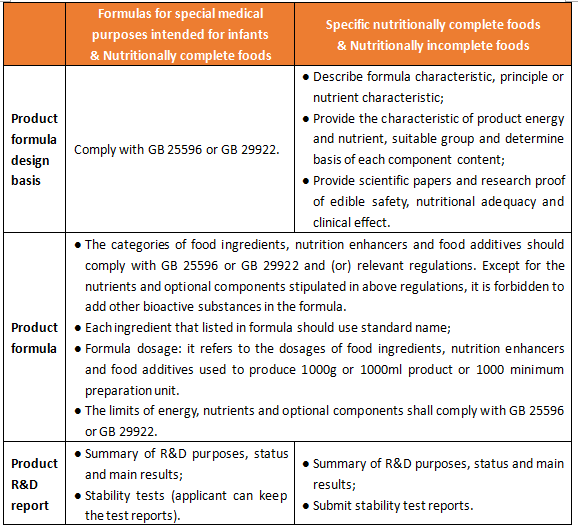
2.2 Products R&D reports, formula materials and formula design basis
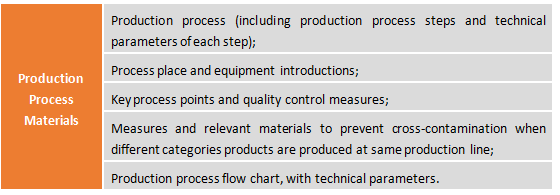
2.3 Production Process Materials
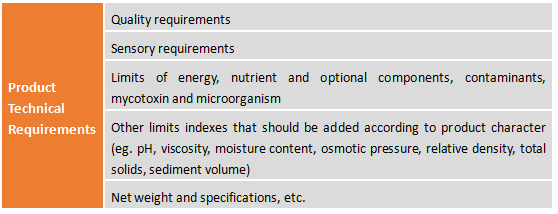
2.4 Product Standard Requirements
Contents of product standard requirements include:
- Overview materials (title page, content, foreword)
- General materials (title, scope, referenced documents)
- Technical materials (technical requirements, test methods, test rules, sign, packaging, storage and annex)
- Quality requirements writing instructions, etc.
Among them, the contents of technical requirements including:
2.5 Samples of Product Label and Instruction Book
Former CFDA had issued the Label and Instruction Book Manuscripts Requirements of FSMP (Trial), and CIRS have analyzed the requirements and made a conclusion. Please click here for further information.
2.6 Test Reports

2.7 Evidence Material of R&D Capacity, Production Capacity and Test Capacity
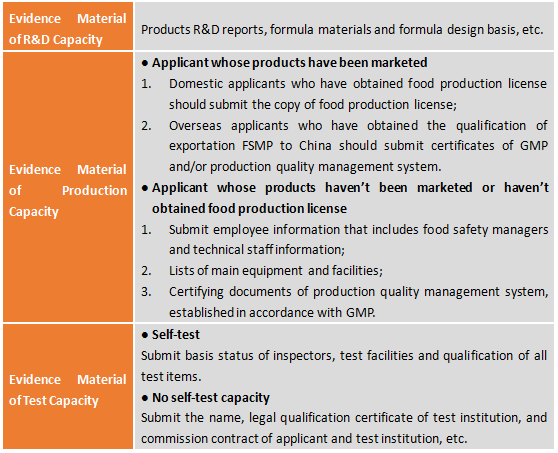
2.8 Clinical Trial Reports (Only Required for Specific nutritionally complete foods)
- The clinical trial report shall be issued by a qualified clinical trial institution entrusted by applicant, and should include a complete statistical analysis report and data;
- Relevant materials of clinical trial, e.g., domestic and/or foreign review materials of clinical trial, etc.
2.9 Other Materials
Certificates related to the registration application, e.g., copy of trademark registration certificates, etc.
For imported products, there are additional documents required:

If you would like to know more information about FSMP registration, please kindly click Foods for Special Medical Registration in China.
If you have any other needs or questions, please feel free to contact us at service@cirs-group.com.
References:
Dossier Requirements of Foods for Special Medical Purpose (Trial) (2017 revision)
