News updates
On June 28, 2023, UK published the REACH (Amendment) Regulations 2023 (No.722) which extends the legislative deadlines for registrants to submit information by 3 years. The REACH (Amendment) Regulations shall enter into force on July 19, 2023. That is to say, from July 19, 2023, the dossier submission deadlines of different tonnage shall be extended to October 27 2026, October 27 2028, and October 27 2030 respectively (new transitional period).
With the first UK REACH submission deadline (October 2023) approaching, the UK government has released a consultation on extending the current submission deadlines. According to the consultation results published on November 29, 2022, 82% of respondents selected to extend the deadline by three years.
There were 20 questions in this consultation document. The extension of the UK REACH submission deadline being in the spotlight was provided with the following alternatives:
- Option 1: extend all the current submission deadlines of each tonnage band by three years to October 2026, October 2028, and October 2030;
- Option 2: extend the first submission deadline by three years to October 2026, the second by two years to October 2027, and the third by two years to October 2028;
- Option 3: do nothing and do not change the current submission deadlines (October 27, 2023; October 27, 2025; and October 27, 2027.)
- Option 4: Do not have a preferred option.
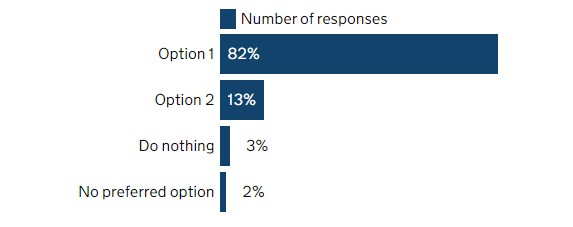
Figure 1: Response rate to this consultation question
Results show that 82% of respondents selected Option 1, which means submission deadlines would be extended to October 2026, October 2028, and October 2030.
The UK government suggested that the extra time under Option 1 could lessen the burden on downstream users and SMEs. Moreover, with the consent of the Scottish and Welsh governments, the UK government will be legislating to extend the current deadlines by three years for each tonnage band.
The UK government will also legislate to extend the statutory dates for compliance checks so that they do not precede the date submission dates.
About UK REACH
The UK REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) Regulation is one of the main pieces of legislation for the regulation of chemicals in Great Britain, which regulates the use of substances in Great Britain, while EU REACH continues to apply in Northern Ireland.
UK REACH requires substances that are manufactured in, or imported into, Great Britain to be registered with the Agency for UK REACH – the Health and Safety Executive (HSE). Registrations include information on the hazards, uses, and exposure of the substance. Registration information is used by the HSE for regulatory purposes and by the registrants to identify appropriate risk management measures for themselves and other users down the supply chain.
The UK REACH Regulation contains transitional provisions to reduce the disruption to the industry as they moved to the new regime from EU REACH. These provisions allow companies to submit initial “notification” data to continue trading and then provide full registration data. The current deadlines for completing this transitional registration process are:
- October 2023 for substances included on the EU REACH candidate list before UK REACH came into effect; substances that are carcinogenic, mutagenic or toxic for reproduction and manufactured or imported in quantities of one tonne a year or more; substances that are very toxic to aquatic life and manufactured or imported in quantities of 100 tonnes or more a year; and all substances manufactured or imported in quantities of 1,000 tonnes or more a year;
- October 2025 for substances added to the UK REACH candidate list before the 2023 submission deadline, and all substances manufactured or imported in quantities of 100 tonnes or more a year;
- October 2027 for all substances manufactured or imported in quantities of one tonne or more a year.
CIRS Comments
These consultation results suggest that Great Britain is significantly likely to have a three-year extension on downstream user import notification (DUIN) and new registration of an existing substance (NRES), which enables enterprises to have sufficient time to make full preparation for dossiers and data. The three-year extension could reduce the unnecessary expenses of enterprises to meet deadlines and data requirements and thus leave them plenty of time to make decisions. CIRS Group will stay focused on this and inform you of the latest updates.
If you need any assistance or have any questions, please get in touch with us via service@cirs-group.com.
Further Information
Consultation Outcome: Summary of Responses and Government Response
The Latest UK REACH Update - DUIN Update and the NRES Process

