Under the Regulations concerning the Hygiene Supervision over Cosmetics, a total of 14 new raw materials were approved from 2004 to May 1, 2021. From May 1, 2021, registration and filing of new cosmetic ingredients under the CSAR shall be applied through the cosmetics declaration and review system under the NMPA official website. As of July 22, 2022, a total of 23 new cosmetics ingredients were filed. Among them, 16 are domestic new cosmetics ingredients, and 7 are imported new cosmetics ingredients.
In July 2022, CIRS Group obtained a filing number of imported new cosmetics ingredients for clients. Earlier in December 2021, CIRS Group helped clients obtain the first and second registration number for imported new cosmetic ingredients.
Detailed information of the new cosmetic ingredients approved for filing are as follows:
Table 1 Filing info of domestic new cosmetic ingredients
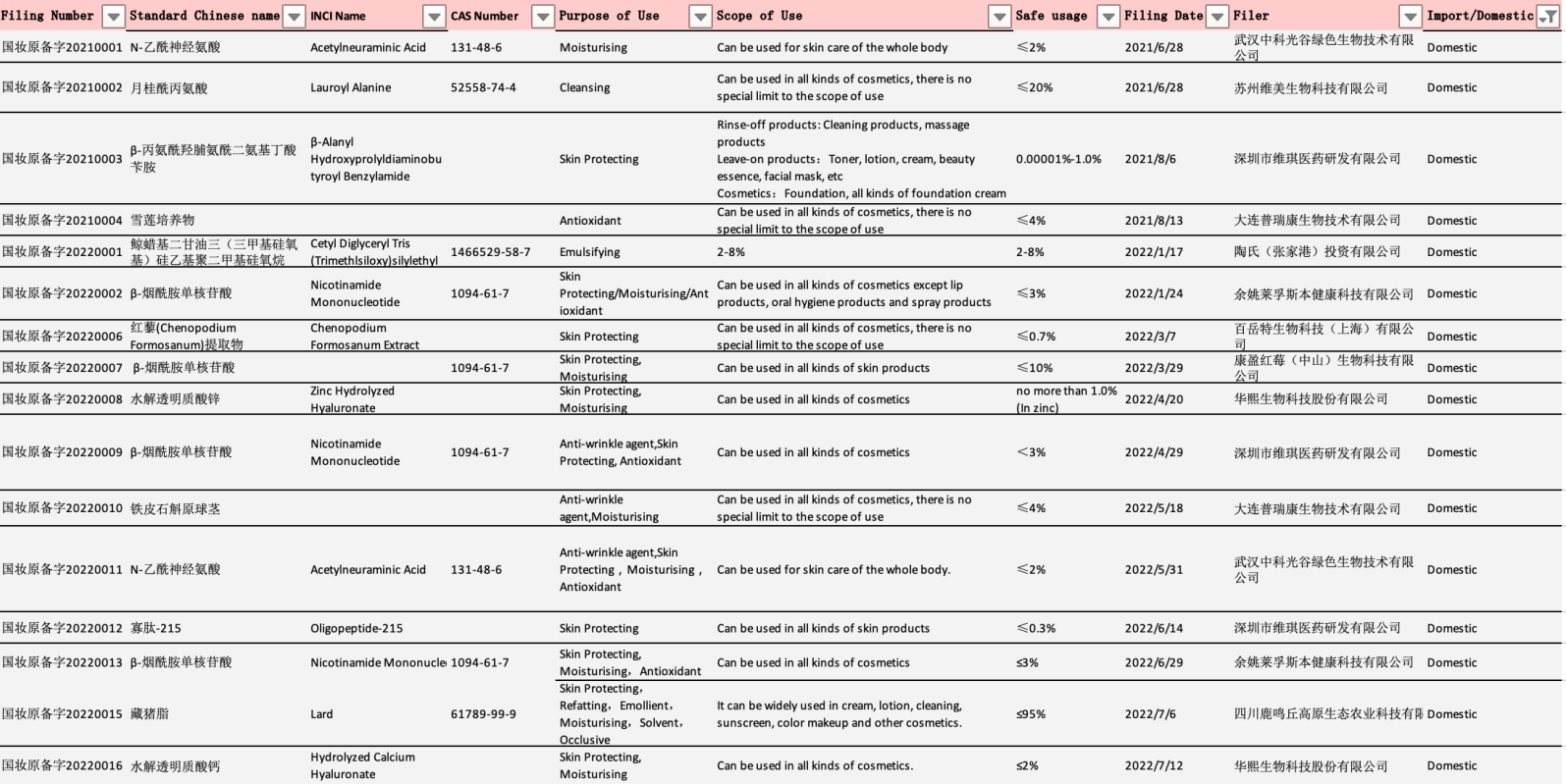
Data Source: NMPA
Table 2 Filing info of imported new cosmetic ingredients

Data Source: NMPA
If you have any needs or questions, please contact us at service@cirs-group.com.

