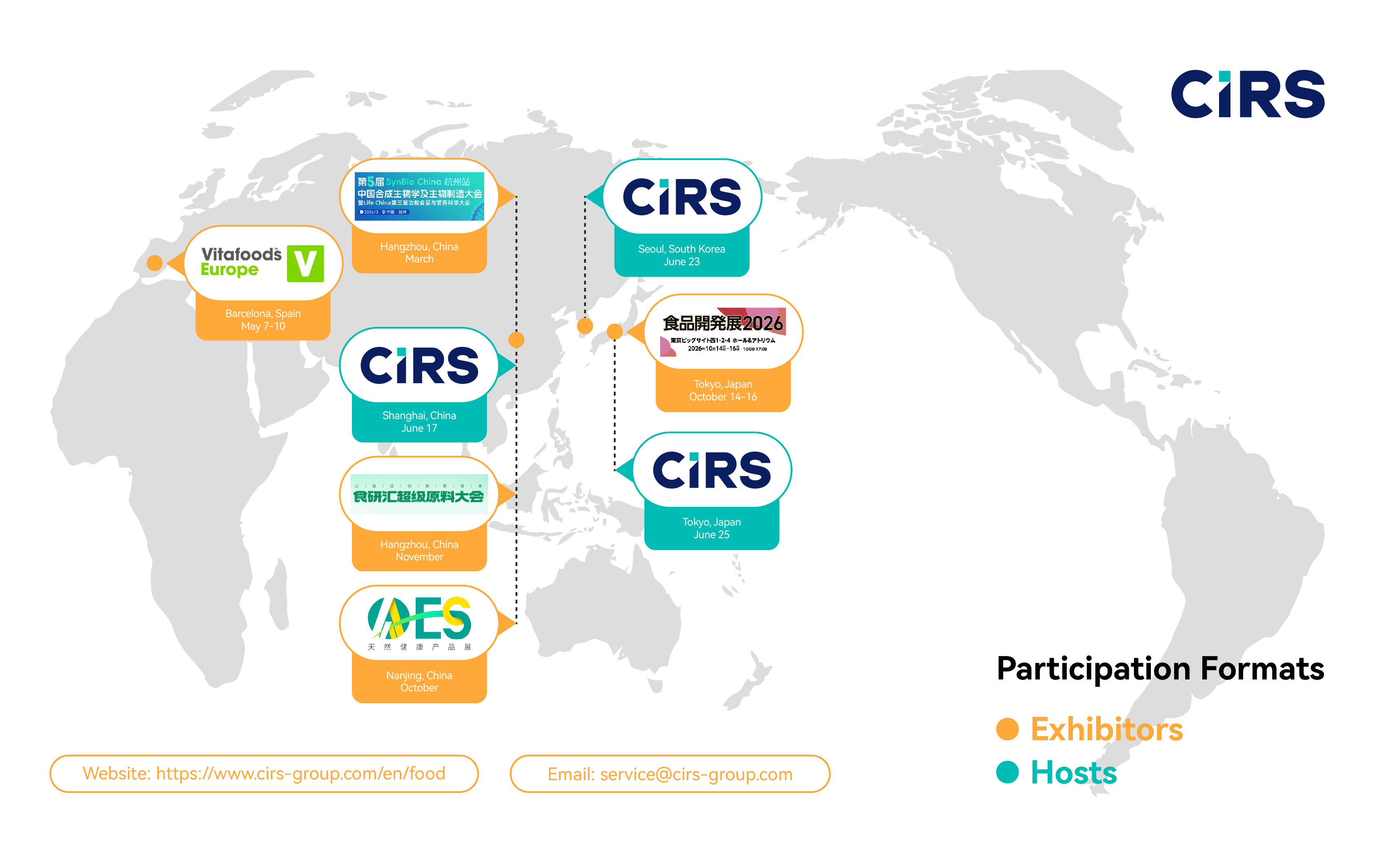
Meet our global food regulatory team at leading 2026 industry events worldwide. Explore our events calendar and book a meeting.



Meet our global food regulatory team at leading 2026 industry events worldwide. Explore our events calendar and book a meeting.

CIRS Group will host the “2026 Global Food Regulations Annual Forum” in Shanghai, Seoul and Tokyo in June 2026. This conference will bring together leading experts from China, the US, Europe, Japan and South Korea to provide a comprehensive overview of the latest global regulatory developments.

CIRS EU, the European subsidiary of CIRS Group, will be exhibiting at Vitafoods Europe in Barcelona, Spain, from May 5–7, 2026 (Booth No.: 6D84M, Hall 6) . CIRS has participated in Vitafoods Europe multiple times in Geneva, and attended the 2025 event in Barcelona. This year, we will once again be present at Vitafoods Europe 2026. We sincerely invite both new and existing partners to join us for discussions and to explore collaboration opportunities together. We look forward to seeing you there.
On April 23, the China State Administration for Market Regulation issued a notice on “Deepening the Governance of the Internet Advertising Ecosystem” and simultaneously released the “Key Tasks for Regulating the Order of the Internet Advertising Market”. The rectification work will last for six months starting from the date of publication.

To help companies better understand the design requirements for the labeling of prepackaged special dietary foods, CIRS Group has compiled a summary of the key changes in this draft for reference, based on the draft and its compilation notes.

On January 27, 2026, Taiwan released the “Guidelines for Safety Assessment of Novel Food Ingredients”, which clarify the definition, required submission materials, and regulatory procedures for novel food ingredients in Taiwan. The guidelines also specify documentation requirements for ingredients produced using genetically modified microorganisms (GMMs), ensuring the safety of such food ingredients for consumption.

On April 22, 2026, China National Center for Food Safety Risk Assessment (CFSA) issued 4 new food materials. Comments are welcomed before May 22, 2026.

On April 17, 2026, the National Center for Food Safety Risk Assessment solicited public comments on expanding the scope of use for the food additive Carmine and the food nutrient enhancer 3'-sialyllactose sodium salt.

Human milk oligosaccharides (HMOs) are the third most abundant solid nutrient in breast milk, following only lactose and lipids. HMOs form a variety of complex structures through different combinations of five basic components—glucose, galactose, N-acetylglucosamine, L-fucose, and N-acetylneuraminic acid—and possess multiple functions, including regulating the gut microbiota, establishing the intestinal barrier, boosting immunity, and enhancing cognitive development.

As of March 31, 2026, a total of 20 Foods for Special Medical Purposes (FSMPs) were approved in the first quarter of 2026, bringing the cumulative number of approved FSMPs in China to 310.